Cercaria caribbea LVIII Cable, 1963 (Digenea: Cyathocotylidae) in the Republic of Korea and Its Surface Ultrastructure
Article information
Abstract
Cercaria caribbea LVIII Cable, 1963 (Digenea: Cyathocotylidae) was detected from a brackish water gastropod species (Cerithideopsilla cingulata) in a coatal area of Shinan-gun, Jeollanam-do (Province), the Republic of Korea, and its surface ultrastructure was studied using a scanning electron microscope. The cercariae were found freely swimming or enveloped within daughter sporocysts when the snail host was mechanically broken. They were morphologically characterized by a linguiform and ventrally concave body, a long and bifurcated tail, and the presence of a holdfast (=tribocytic) organ posterior to the ventral sucker. On the whole ventral and dorsal surfaces, peg-like tegumental spines were densely distributed. Around the oral sucker, several sensory papillae, each with a short cilium, were distributed, and on the tail, sensory papillae, each with an extensively long cilium, were observed. This is the first record describing a cyathocotylid cercaria from a brackish water gastropod in the Republic of Korea.
In the trematode life cycle, snails have an important role of the first, or first and second, intermediate host [1]. In the Family Cyathocotylidae Muhling, 1898, the final hosts include reptiles, birds, and mammals. Mother and daughter sporocysts are found in gastropods, and metacercariae are detected in fishes, amphibians, and aquatic invertebrates [2]. Little has been known about the kinds of cyathococylid cercariae in marine gastropods in the Republic of Korea. In the present study, we detected sporocysts and cercariae of Cercaria caribbea LVIII Cable, 1963 in a gastropod snail, Cerithideopsilla cingulata, from Shinan-gun, Jeollanam-do (Province), the Republic of Korea, and their surface ultrastructure was studied.
The gastropod snails, C. cingulata (2.3×0.9 cm in length and diameter), were collected from a coastal area of Shinan-gun, Jeollanam-do, the Republic of Korea. To obtain freshly emerging cercariae, the snails were individually placed in a small vial containing filtered seawater and left at room temperature until examined using a dissecting microscope. Daughter sporocysts (hereafter sporocysts) were obtained by dissecting infected gastropods. The larval stages were studied live, unstained, or vitally stained with 5% neutral red, and also after fixation with hot acetic acid-formalin-alcohol fixative and staining with aceto-carmine. Measurements, in micrometers, were done on 20 vitally stained specimens, with the range and mean in parentheses.
The cercariae were washed with 0.2 M cacodylate buffer (pH 7.3) and fixed with 2.5% glutaraldehyde solution. They were dehydrated with a graded series of ethanol, dried in a critical point dryer, mounted on aluminum stubs, and coated with gold using an ion sputtering coater (IB-3, Giko Engineering Co., Tokyo, Japan). The specimens were observed using a scanning electron microscope (ISI DS-130C, Akashi Co., Tokyo, Japan) at an accelerating voltage of 10 kV.
Seven C. cingulata (0.3%) were found infected with sporocysts of C. caribbea LVIII out of a total of 2,015 snails examined. As many as 76-131 sporocysts (13.6±0.9×0.8±0.03 mm) were found per snail, and 310 to 394 (mean 353) cercariae (160±33×105±18 µm) were detected per snail (Table 1).
Description of sporocysts and cercariae is based on light and scanning electron microscopic examinations. Sporocysts had a cylindrical body with transverse ridges on the tegumental surface (Fig. 1A). Mature and immature cercariae were observed within the sporocysts (Fig. 1B, C). The cercarial body was linguiform, concave ventrally, and its tail was bifurcated with characters of the cyathocotylid group (Figs. 1D, E, 2A). The tegument of the body and tail was covered with densely distributed sharp-pointed peg-like spines (Fig. 2B-J). Single type I papillae were circularly arranged on the ventral surface of the body symmetrically.
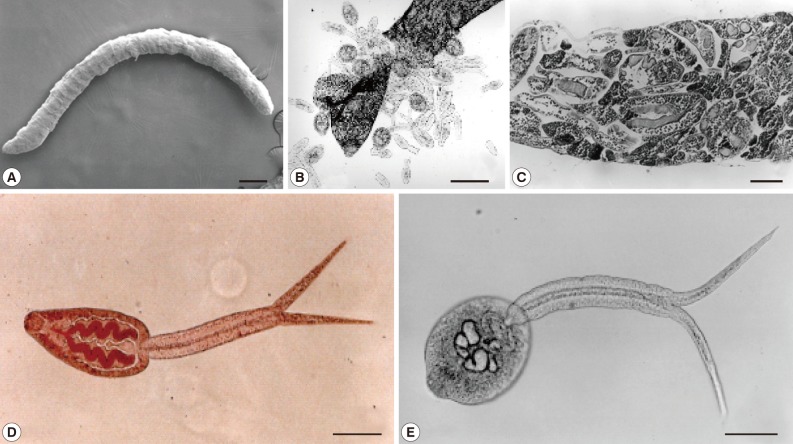
Morphology of a sporocyst (A) and cercariae (B-E) of Cercaria caribbea LVIII from a brackish water gastropod, Cerithideopsilla cingulata, in Korea. (A) A whole body scanning electron micrograph of a sporocyst. Bar=220 µm. (B) Mature and immature cercariae with cercarial embrios are escaping from the ruptured birthpore of a sporocyst. Bar=160 µm. (C) Cross-section of a sporocyst containing cercariae. Bar=25 µm. (D) A cercaria vitally stained with neutral red. Two convoluting ceca are seen as red color. Bar=25 µm. (E) An unstained cercaria. Bar=25 µm.
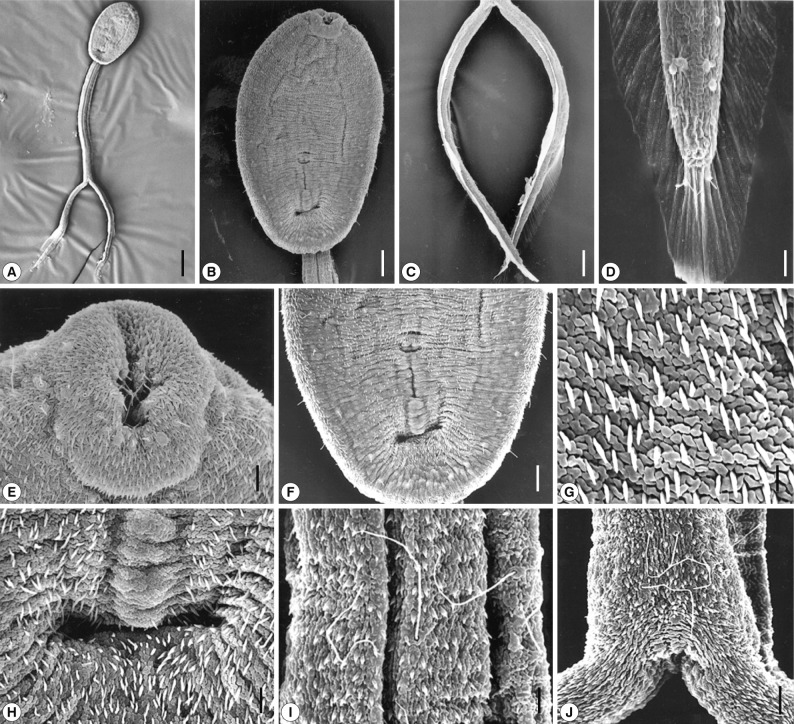
Scanning electron micrographs of Cercaria caribbea LVIII cercaria. (A) Ventral view of a cercarial body and bifurcated tail. Bar=50 µm. (B) A cercarial body showing the oral and ventral suckers, ventral view. Tegumental spines are densely distributed in anterior surface of the body. Bar=14 µm. (C) The bifurcated tails look like oars. Bar=17 µm. (D) On the tip of each furca, several spines are observed, which are larger than those of the general cuticular spination. Two sensory papillae with a long cilium are seen on the fin (lower) and 2 others are obsercved anterior to the tip (upper). Bar=3 µm. (E) On the lip of the oral sucker, sensory papillae with a short cilium are distributed, and peg-like spines are seen on the surface. Bar=3 µm. (F) Ventral sucker was premature, and a primordial tribocytic organ is seen posteriorly close to the ventral sucker. Bar=8 µm. (G) Peg-like tegumental spines densely distributed on the body surface and furcae. Bar=1 µm. (H) A tegumental groove or pit located posteriorly far from the tribocytic organ. Bar=3 µm. (I) Surface of a part of the tail stem revealing sensory papillae with a long cilium. Bar=3 µm. (J) Long-ciliated sensory papillae on the base of the furcae. Bar=3 µm.
The oral sucker was small and subterminal. Several grouped type I papillae with a short cilium were observed on the lip of the oral sucker. Several type I papillae were also found around the oral sucker (Fig. 2E). The ventral sucker was embryonic. In a short distance posterior to the ventral sucker, the primordium of the tribocytic organ was observed. The tegument posterior to the primitive tribocytic organ was wrinkled, with a deep central groove and numerous centrifugally arranged shallow grooves (Fig. 2H). On the bifurcated tail, short spines with a broad base and a single pointed tip were densely distributed (Fig. 2I, J). Two papillae with a long cilium were observed on the fin, and 2 other papillae were observed near the tip of the tail (Fig. 2D). From the base of the tail tip, the finely ribbed fin was observed continuously along the dorsal and ventral edges of each furca. On the tip of each furca, several spines were observed which were larger than those of the general cuticular spination (Fig. 2C, D).
Based on the above characters, we identified the sporocysts and cercariae found in the present study as C. caribbea LVIII Cable 1963 [3]. Although the size of the cercariae was about 2/3 of the original description, other morphological characters were well corresponded with the descriptions of C. caribbea [3]. Cyathocotylid trematodes have great morphological variability in their adult and metacercarial stages [2]. However, the morphology of their cercariae is highly homogenous [3]. For this reason, the cercarial stage of cyathocotylids has advantages over adults in investigating the zoogeography of these trematodes, and correct identification is essential for such studies.
The numbering of C. caribbea series was initiated by Cable in 1956 with reporting 51 species from marine areas of Puerto Rico [3]. The 52nd species, C. caribbea LII (probably Haplodena varia Haplosporidae), was described in 1962 from Gulf-Carribean regions [4]. Subsequently, the 53rd to 74th species were added from marine areas of Curaçao and Jamaica in 1963 [3]. However, the life cycle of the majority of the species is yet unknown. C. caribbea LVIII was originally described in a gastropod Cerithium variabile from Jamaica [3]. After then, little has been known about this cercaria. Although size variation existed between our study and the previous report [3], other morphological characters of potential taxonomic importance in our specimens were identical to those of the original report. There were 2 cyathocotylid cercariae, C. caribbea LI and C. carribea LVIII, among those reported [3], and our specimens were consistent with the latter species.
Sensory papillae with a long cilium were observed on the body and tail of various fluke species; Neodiplostomum seoulense [5], Clonorchis sinensis [6], and Metagonimus yokogawai [7]. The sensory papillae are thought to be essential for the cercariae to find, attach, and penetrate into the fish host because they are not found on the metacercariae of those flukes. However, our cyathocotylid cercariae, C. carribea VLIII, had sensory papillae with an extremely long tail, which is characteristic from other cercarial species.
Cercaria carribea XXXI, a microphallid species, was recently found from an esturine snail, using crabs as second intermediate hosts in mangrove areas of Puerto Rico [8]. In Korea, at present, only C. carribea LVIII among these cercarial series has been found. The presence of C. carribea LVIII in Jamaica, near the Atlantic Ocean, and in Korea, near the Pacific Ocean, may indicate a geographically wide distribution of this trematode species.
To date, the relationships of cyathocotylid groups of cercariae in the systematics of the family Cyathocotylidae are unclear. Thus, further studies are required on the life cycle and systematics of the cyathocotylid cercariae.
