Albendazole and Mebendazole as Anti-Parasitic and Anti-Cancer Agents: an Update
Article information
Abstract
The use of albendazole and mebendazole, i.e., benzimidazole broad-spectrum anthelmintics, in treatment of parasitic infections, as well as cancers, is briefly reviewed. These drugs are known to block the microtubule systems of parasites and mammalian cells leading to inhibition of glucose uptake and transport and finally cell death. Eventually they exhibit ovicidal, larvicidal, and vermicidal effects on parasites, and tumoricidal effects on hosts. Albendazole and mebendazole are most frequently prescribed for treatment of intestinal nematode infections (ascariasis, hookworm infections, trichuriasis, strongyloidiasis, and enterobiasis) and can also be used for intestinal tapeworm infections (taeniases and hymenolepiasis). However, these drugs also exhibit considerable therapeutic effects against tissue nematode/cestode infections (visceral, ocular, neural, and cutaneous larva migrans, anisakiasis, trichinosis, hepatic and intestinal capillariasis, angiostrongyliasis, gnathostomiasis, gongylonemiasis, thelaziasis, dracunculiasis, cerebral and subcutaneous cysticercosis, and echinococcosis). Albendazole is also used for treatment of filarial infections (lymphatic filariasis, onchocerciasis, loiasis, mansonellosis, and dirofilariasis) alone or in combination with other drugs, such as ivermectin or diethylcarbamazine. Albendazole was tried even for treatment of trematode (fascioliasis, clonorchiasis, opisthorchiasis, and intestinal fluke infections) and protozoan infections (giardiasis, vaginal trichomoniasis, cryptosporidiosis, and microsporidiosis). These drugs are generally safe with few side effects; however, when they are used for prolonged time (>14–28 days) or even only 1 time, liver toxicity and other side reactions may occur. In hookworms, Trichuris trichiura, possibly Ascaris lumbricoides, Wuchereria bancrofti, and Giardia sp., there are emerging issues of drug resistance. It is of particular note that albendazole and mebendazole have been repositioned as promising anti-cancer drugs. These drugs have been shown to be active in vitro and in vivo (animals) against liver, lung, ovary, prostate, colorectal, breast, head and neck cancers, and melanoma. Two clinical reports for albendazole and 2 case reports for mebendazole have revealed promising effects of these drugs in human patients having variable types of cancers. However, because of the toxicity of albendazole, for example, neutropenia due to myelosuppression, if high doses are used for a prolonged time, mebendazole is currently more popularly used than albendazole in anti-cancer clinical trials.
INTRODUCTION
Albendazole, methyl [5-(propylthio)-1H-benzimidazol-2-yl] carbamate, is a broad- spectrum anti-parasitic agent, first introduced in 1975 for treatment of liver flukes, tapeworms, lung and gastrointestinal nematodes in sheep and cattle [1]. It was subsequently approved for human use in 1982 [2,3]. Mebendazole, methyl 5-benzoyl-1H-benzimidazol-2-yl-carbamate, is also a broad-spectrum anthelmintic, applied first to human subjects in 1971 [4]. Both drugs are relatively insoluble in water and most organic solvents and poorly absorbed from the gastrointestinal tract unless administered with high-fat meals [5]. The absorption rate of oral albendazole and mebendazole in human intestine is about 1–5% [3]. It is assumed that these drugs act directly on luminal parasites in the gastrointestinal tract, and after first-pass in the intestinal wall and liver their metabolites are active against parasites in internal organs and tissues [3,4]. For example, albendazole sulfoxide, among 5 metabolites of albendazole detected in human urine, is considered the major metabolite responsible for the anthelmintic activity [3,6]. In mebendazole, 2 forms of metabolites are known in the plasma, i.e., aminometabolite and hydroxymetabolite, which are excreted in urine as decarboxylated forms [6]. Albendazole and mebendazole are known to block microtubule functions of parasites and mammalian cells through inhibition of polymerization of ß-tubulin into microtubules followed by inhibition of glucose uptake and transport which eventually lead parasites to be in shortage of glycogen [7].
Since albendazole and mebendazole were first used for treatment of several intestinal helminth species infecting humans [2,4], numerous studies have been performed on their anthelmintic efficacies against a variety of nematodes, trematodes, cestodes, and even protozoans [1,3,5–12]. The examples of nematode infections included ascariasis, hookworm infections, trichuriasis, strongyloidiasis, enterobiasis, larva migrans (visceral, ocular, neural, and cutaneous), anisakiasis (anisakidosis), trichinosis, angiostrongyliasis (cerebral and abdominal), gnathostomiasis, capillariasis (hepatic and intestinal), thelaziasis, lymphatic filariasis, onchocerciasis, and dirofilariasis [8,9]. Trematode infections that can be treated with albendazole and/or mebendazole are fascioliasis, clonorchiasis, opisthorchiasis, and intestinal fluke infections [13–20]. Cestode diseases responsive to albendazole and/or mebendazole include cysticercosis, hydatidosis, and intestinal taeniases [21–25]. Protozoan diseases for which albendazole and/or mebendazole have been tried were giardiasis, vaginal trichomoniasis, cryptosporidiosis, and microsporidiosis, although the efficacies against some of these infections need to be further evaluated [12,26–31].
Meanwhile, albendazole and mebendazole have been shown to suppress tumor growth in vitro and in vivo and thus have been repositioned as anti-cancer agents [32–35]. For example, albendazole induces oxidative stress promoting DNA fragmentation and triggering apoptosis and inducing death of breast cancer cells [33]. Mebendazole disrupts microtubule functions by preventing polymerization of tubulin in cancer cells, such as glioblastoma and melanoma, and leads to cell death [32]. In addition, mebendazole would synergize with a range of other anti-cancer drugs, including the existing ones [32].
Albendazole and mebendazole are generally safe without significant side effects when used in recommended doses just for 1–3 days [2,4,9,13]. However, when these drugs are used for prolonged periods to treat tissue helminthiases [36], cancers [37,38], or even when they are used just for once as preventive chemotherapy for intestinal helminths [39], liver toxicity, allergic reactions, and rarely severe myelosuppression (in particular, neutropenia) may occur. In addition, the occurrence of drug resistance to albendazole and mebendazole with treatment failure was reported recently in hookworm infections, trichuriasis, ascariasis, and lymphatic filariasis [40–42] which has become a significant emerging issue.
The present study aimed to review briefly recent trends in use of albendazole and mebendazole not only as anti-parasitic agents but also as new promising anti-cancer agents. In addition, the increasing reports on the hepatotoxicity of these drugs as well as drug resistance in parasites are also briefly reviewed.
INTESTINAL HELMINTHIASES
Ascariasis
Among various species of intestinal nematodes infecting humans, Ascaris lumbricoides is the most highly susceptible parasite species to albendazole and mebendazole [1,8–10,42–44] (Table 1). The earliest clinical study in 1982 using albendazole (400 mg in a single dose) reported the cure rate (CR) (=egg negative conversion rate; NCR) to be 96%, and the egg reduction rate (ERR) almost 100% [2]. A reduced dose, i.e., 200 mg in a single dose, was also found to be equally effective against A. lumbricoides infection showing 92.3% CR and 98.3% ERR [8]. However, viewing from the drug efficacy and safety, the recommended dose of albendazole for A. lumbricoides has been set at 400 mg single dose by World Health Organization (WHO) [45]. The therapeutic efficacy of albendazole against A. lumbricoides seems unchanged until recently (2017); it was reported to be 95.7–96.5% in CR and 98.5–99.7% in ERR [42,43]. Albendazole (400 mg single dose) was shown to be useful for short-term control (4 months) of ascariasis using mass drug administration (MDA) in schoolchildren in Yangon Region, Myanmar [46].

Use of albendazole and mebendazole for treatment of intestinal helminth infections (nematodes and cestodes)
The effect of mebendazole against intestinal nematodes, including A. lumbricoides, was reported at an earlier time in 1971 from schoolchildren in Brazil using the dose of 100 or 200 mg twice daily for 3–4 consecutive days or 100 mg daily for 6 days [47]. The cure rate for ascariasis was 100% [47]. However, these regimens (repeated doses for 3–6 days) were inconvenient especially for MDA. Therefore, reduced single doses were tried by various researchers [48–50]. The study using a 200 or 300 mg single dose in Costa Rica reported 93–100% cure rates of A. lumbricoides infection and almost 100% ERR [48]. Another study in South Korea reported that a single dose of 200–300 mg revealed 93.8–96.7% CR and 98.6–99.9% ERR [49]. A lower dose, 100 mg once, was almost equally effective as 200–300 mg, showing 89.7% CR and 98.6% ERR [49]. On the other hand, a trial using 500 mg single dose of mebendazole was reported to be highly effective for ascariasis with 93.4% CR and 99.0% ERR [50]. In view of the efficacy and safety, the recommended dose of mebendazole for ascariasis has been set at 500 mg single dose by WHO [45]. The therapeutic efficacy of mebendazole against A. lumbricoides seems unchanged until recently (2017); it was reported to be 96.2% in CR and 98.0% in ERR [43].
Hookworm infections
The use of albendazole for treatment of hookworm infections in humans, i.e., Ancylostoma duodenale and Necator americanus in most cases, started in the early 1980s [2,51,52]. Pene et al. [2,51] reported that the CR and ERR using 400 mg single dose were 80.7–94.3% and 97.0%, respectively, whereas Soula and Siopathis [52] reported lower effects, 74.0% and 97.5%, respectively. Thereafter, until March 1999, at least 68 articles reported the efficacy of albendazole (400 mg single dose; the recommended dose by WHO [45]) against hookworm infections (A. duodenale or N. americanus), with CR ranging from 33.3 to 100% (av. 77.7%), and ERR from 29.6 to 100% [53]. The efficacy was markedly different between the species of hookworms and between adults and children age groups. The average CR for A. duodenale was higher (91.8%) than that for N. americanus (75.0%), and those for adults and children in A. duodenale were similar, 91.7% and 90.8%, respectively, whereas those for adults and children in N. americanus were 80.9% and 67.0%, respectively [53]. These results represent lower efficacy of albendazole in treatment of N. americanus hookworm infections especially in children. Similar efficacies were reported after 2000, for example, Keiser and Utzinger [10] reported that the CR of albendazole for hookworms using 400 mg single dose was 72.0% and the ERR was 64.0–100% (Table 1), and Moser et al. [43] reported 79.5% CR and 89.6% ERR.
Mebendazole showed poor efficacy for hookworms if used 500 mg only 1 time. Keiser and Utzinger [10] and Soukhathammavong et al. [44] reported the CRs of 500 mg single dose of mebendazole to be 15.0% and 17.6% and ERR 0–98.0% and 76.3%, respectively. In comparison, 100 mg twice daily for 3 days (total 600 mg), the recommended dose by WHO [45], showed fairly higher efficacies of 80.0% CR and 41.0–100% ERR according to reports from 27 trials [10]. However, the efficacy of mebendazole was variable depending on the species of hookworms. For example, using the recommended dose of mebendazole, the CR of A. duodenale was 71.4% and the ERR was 97.3% as reported by Seo et al. [54], whereas the CR of N. americanus was lower 52.0% and the ERR was 91.2% as reported by Sinniah et al. [55].
Tribendimidine (400 mg single dose) may be an alternative drug to treat hookworm infections having CR of 83.4% (68.7–92.0%) and ERR of 91.6% (82.6–96.8%) [42].
Trichuriasis
The efficacy of albendazole for Trichuris trichiura was first studied in the early 1980s [2,51,52,56]. Some of these studies reported quite satisfactory results using 200 mg twice (400 mg in total) with CR of 80.0–88.0% and ERR of 98.0–99.0% [51,56]. However, some others using 400 mg in a single dose or 200 mg twice reported lower efficacies, 50.0–61.5% in CR and 76.5–95.0% in ERR [2,13,52]. WHO [45] recommended 400 mg single dose as the recommended dose for moderate T. trichiura infection, but in heavy infections 3-day course (total 1,200 mg) has been recommended. Horton [53] summarized the effects of albendazole (400 mg single dose) for T. trichiura reported until March 1998 worldwide (58 studies) to be highly variable, from 4.9% to 100% (av. 47.7%) in CR and 28.0% to 99.0% (av. 75.4%) in ERR. However, the therapeutic efficacy of albendazole against trichuriasis seemed to be progressively lowered. A study in Lao PDR in 2012 reported only 33.3% CR and 67.0% ERR [44]. In 2 publications in 2017 [43] and 2019 [42], the CRs were reported as 30.7% and 32.1%, and the ERRs were 49.9% and 64.3%, respectively. Moser et al. [42,43] put an emphasis on a temporal decrease of ERR which was 72.6% in 1995 and 43.4% in 2015 and considered this temporal decrease of albendazole efficacy against T. trichiura to be in part due to an emergence of drug resistance. Chai et al. [46] tried to control A. lumbricoides and T. trichiura infections among schoolchildren in Myanmar by MDA using a 400 mg single dose of albendazole but difficulties were experienced in short-term control of trichuriasis seemingly because of limited drug efficacy against T. trichiura.
Mebendazole was first tried to treat T. trichiura infection in the early 1970s [48]. A single dose of 300 mg or 200 mg showed 44.4% or 70.0% CR and 96.4% and 98.3% ERR, respectively, and 100 mg b.i.d. for 2–4 days increased the efficacies, 75.0–90.0% in CR and 99.3–99.6% in ERR [48]. However, Seo et al. [54] obtained a poor efficacy of 100 mg mebendazole twice daily for 3 days against T. trichiura infection only as 27.3% CR and 65.5% ERR. Kim [57] used 2 dosage regimens (total 600 mg), 100 mg b.i.d. for 3 days and 300 mg daily for 2 days, and obtained different results, 36.1% CR and 79.3% ERR for the former and 71.0% CR and 85.4% ERR for the latter regimen. Abadi [50] reported fairly good results using 500 mg single dose of mebendazole, 77.6% CR and 92.8% ERR. WHO [45] set 100 mg b.i.d. for 3 days as the recommended dose of mebendazole for treatment of T. trichiura infection with a 500 mg single dose as an alternative regimen. It is of note that Seo et al. [58] reported a satisfactory effect in long-term control of T. trichiura infection in Korea by repeated MDA using the recommended 3-day regimen of mebendazole, from 40.0% pre-control prevalence to 5.6% post-control prevalence a year later. Recently, however, the 500 mg single dose regimen showed lower efficacies, 27.9% CR and 66.0% ERR in 2012 [44] (Table 1), 42.1% CR and 66.0% ERR in 2017 [43], and 44.4% CR and 80.7% ERR in 2017 [42]. Moser et al. [42,43] reported a more remarkable (compared with albendazole) temporal decrease of ERR in T. trichiura which was 91.4% in 1995 that became 54.7% in 2015. They considered this temporal decrease of the drug efficacy to be in part due to an emergence of drug resistance of worms.
Oxantel pamoate (20 mg/kg single dose) alone or in combination with albendazole (400 mg single dose) can be an alternative drug or drug regimen for trichuriasis [42].
Strongyloidiasis
Albendazole was evaluated to be partially effective for treatment of Strongyloides stercoralis infection, whereas mebendazole was never shown to be effective for strongyloidiasis [59]. In 1982, albendazole (400 mg daily for 3 days) was reported to have some effects against S. stercoralis infection, 48.0% in CR [51]. The ERR for this infection was not calculated because the diagnosis of strongyloidiasis is usually based on detection of larvae by a concentration method [53]. In addition, higher doses of albendazole, 400 mg b.i.d. for 3 or 7 days, revealed 100% CR [14] (Table 1). WHO [45] recommended the regimen of albendazole for strongyloidiasis as 400 mg daily for ≥3 days. Horton [53] reviewed the efficacy of albendazole (400 mg daily for 3 days) against S. stercoralis infection, and the average CR was 62.2% (16.7–100% in range) calculated from 19 studies performed until March 1998. However, in immunocompromised patients with S. stercoralis hyperinfection syndrome, even 5 courses of albendazole (400 mg daily for 3 days) failed to cure the disease [60].
An alternative drug, ivermectin (200 μg/kg daily for 2 days), resulted in more people cured than albendazole, and equally tolerated [59]. In northwestern Argentina, albendazole plus ivermectin was tried to control. Stercoralis and hookworm infections and was found to be effective for reduction of the prevalence as well as the morbidity due to these parasite infections [61].
Enterobiasis
Both albendazole and mebendazole have been shown to be highly effective against Enterobius vermicularis infection. The first clinical trial of albendazole was performed in the early 1980s [53]. Thereafter until March 1998, many clinical trials (at least 26 trials) were performed using 400 mg single dose of albendazole (recommended dose by WHO [45]) [53]. In most trials, the CR at day 14–17 post-treatment ranged from 93.3 to 100% [53]. In South Korea, Rim et al. [13] reported 98.1% CR when follow-up examinations were done on day 14 and 28 post-treatment (Table 1). Also, Chai et al. [62] obtained 100% CR at day 14 post-treatment using 400 mg single dose of albendazole. Reduced doses, 100 mg or 200 mg in a single dose, of albendazole were as effective as the recommended dose [63]. For chemotherapeutic control of enterobiasis using MDA with albendazole in a family or a group of people, treatment should be repeated at least 2 or more times every 2–3 weeks [8,45].
Mebendazole was first used in clinical trial for enterobiasis in 1971 [4]. This drug, in a standard dose of 100 mg single dose [45], was reported to have been satisfactory for treatment of E. vermicularis infection in adults (88% CR) as well as children (91%) [4]. Higher doses, 100 mg b.i.d. for 3 days (total 600 mg) or 200 mg or 400 mg in a single dose, revealed similar therapeutic effects [4,54,63]. It is notable that the anthelmintic efficacy of mebendazole against young/juvenile worms of E. vermicularis was higher than that of pyrvinium pamoate or pyrantel pamoate [64]. Thus, the rate and timing of egg positive conversion (due to limited drug efficacy) after MDA were low and delayed in a highly endemic group of children if mebendazole (100 mg single dose) was used but the rate and timing of egg positive conversion were high and rapid if pyrvinium pamoate or pyrantel pamoate was used [64]. For chemotherapeutic control of enterobiasis using mebendazole 100 mg single dose in a group of children, MDA should be repeated at least 2–3 times at 3-week intervals [65].
Taeniases
Albendazole was used for treatment of taeniases due to Taenia spp. (T. saginata and T. solium) for the first time by Rim et al. [13,21]. Whereas single dose regimens (400 mg or 800 mg) were almost ineffective, the dosage showing therapeutic efficacy was 400 mg b.i.d. or t.i.d. for 2–3 days (total 1,600–3,600 mg) [13,21]. Using this dosage regimen, slightly better efficacy was obtained against T. saginata (90.0–95.0% CR) than against T. solium (70.0–92.9% CR) infection [13,21] (Table 1). Thereafter, based on 8 trials performed until March 1998 using a reduced dosage regimen of 400 mg daily for 3 days (total 1,200 mg), CR of 75.7–100% was obtained [53]. In other trials, a single dose of 400 mg for adults and 200 mg for children showed low efficacy with 50.0–58.8% CR, whereas triple doses repeated for 3 days brought about 100% CR [23,66].
Mebendazole was also proved to be effective against Taenia spp. infection if used in multiple doses, for example, 100 mg b.i.d. for 3 days [67], 300 mg b.i.d. for 3 days [68], or 500 mg daily for 3 days [23]. In particular, the CR of multiple doses, 500 mg daily for 3 days, was 100%, whereas that of 500 mg single dose was only 50.0% [23].
Praziquantel (5–10 mg/kg single dose) is the drug of choice for human intestinal taeniases [69]. Niclosamide is an alternative drug.
Hymenolepiasis
The therapeutic efficacy of albendazole against human Hymenolepis nana infection has been variable [53]. Whereas Rossignol and Maisonneuve [70] reported the CR of only 29.4% using 400 mg albendazole daily for 3 days, Rim et al. [13] reported the CR of 66.7% using 800 mg daily for 2 days (Table 1). Horton [53] reviewed the CR of human H. nana infection using 400 mg albendazole daily for 3 days in 13 trials from 1981 until March 1998 to be in the range between 28.5% and 100% (av. 69.5%). However, shorter courses, particularly 400 mg single dose, do not appear to produce a significant cure of the disease [53].
Human hymenolepiasis (H. nana) has been shown to be relatively insensitive to a standard 3-day course regimen of mebendazole (100 mg b.i.d. for 3 days) with CR ranging from only 18% to 40% [71].
Praziquantel (15–25 mg/kg single dose) is the drug of choice for hymenolepiasis [69]. However, to avoid recurrence of hymenolepiasis, a repeated treatment after 14 days is recommended [69].
TISSUE HELMINTHIASES
Visceral, ocular, and neural larva migrans
Human toxocariasis or visceral, ocular, and neural larva migrans are most commonly caused by the larvae of Toxocara canis (dog ascarid) and less commonly Toxocara cati (cat ascarid) [72]. Albendazole was experimentally shown to be effective against the larval stage of T. canis in mice [73]. Subsequently, albendazole began to be used for treatment of human toxocariasis (visceral or ocular larva migrans) with the dosage regimen of 5 mg/kg b.i.d. for 5 days; however, only 6 (31.6%) of 19 patients were clinically cured, and 11 (57.9%) showed excellent or good drug tolerability [74]. In ocular toxocariasis (5 patients involving 7 eyes), treatment with a higher dose for longer days, 800 mg b.i.d. for 14 days, produced good results with no relapse and no side effects [75]. In the United States, the regimen of albendazole to treat visceral larva migrans was set at 800 mg daily for 5 days [76] (Table 2). However, for neural toxocariasis albendazole should be given 800 mg daily (15 mg/kg for children) for longer duration, 21–28 days [77–79]. Moreover, in Japan, even an additional second course of 28-day treatment, following the first 28-day course treatment with albendazole (10–15 mg/kg daily) and 14 days of drug free, was needed to obtain cure of 192 (78.0%) of 246 patients with larva migrans syndrome [80].
Mebendazole was tried to treat visceral larva migrans. At least 20–25 mg/kg daily for 21 days was needed to treat human toxocariasis [81]. However, The Medical Letter [76] recommended 100–200 mg b.i.d. for 5 days for human visceral larva migrans due to Toxocara spp.
Cutaneous larva migrans
Cutaneous larva migrans is most commonly caused by the larvae of Ancylostoma brasiliense (dog and cat hookworm) and less commonly Ancylostoma caninum (dog hookworm). Albendazole at the dose of 400 mg daily for 5 days showed fairly good results in 15 of 18 patients with cutaneous larva migrans [82]. However, longer period use of oral albendazole (400 mg daily), for 10–21 days, was reported in 5 cases of cutaneous larva migrans over buttocks and perianal regions in children [83]. Topical application of 4% albendazole cream (office-made with Vaseline) on the affected skin lesions of 3 cutaneous larva migrans cases also showed effects in 2 adults and 1 child patient [84].
Mebendazole has seldom been tried to treat cutaneous larva migrans because it seems less efficacious than albendazole. A 47-year-old man having erythematous, serpiginous burrows on both feet and right thigh and buttock received 2 courses of mebendazole therapy (3 days each, drug dosage not described), and 70% resolution of skin lesions was obtained [85]. However, he developed pulmonary infiltrates with Löffler syndrome (shortness of breath, wheezing, and chest discomfort), and was prescribed with albendazole and intravenous hydrocortisone for 5 days and then completely cured [85].
Anisakiasis (anisakidosis)
Human anisakiasis (or anisakidosis) is mainly caused by Anisakis simplex, Anisakis pegreffii, Anisakis physeteris, and Pseudoterranova decipiens, and the treatment of choice is gastroendoscopic removal of the larvae or surgical excision of the affected lesion [86,87]. However, albendazole or mebendazole may be prescribed as a supplementary therapy [9,88–90] or in cases where the larvae have penetrated the thickness of the stomach or intestinal wall [91]. The use of albendazole (400 mg b.i.d for 21 days) in human anisakiasis was first reported by Moore et al. [88] which was followed by several workers [89]. However, the use of albendazole alone is controversial regarding its efficacy for treatment of this disease [91].
Mebendazole was rarely tried for treatment of human anisakiasis. Maggi et al. [92] used mebendazole 100 mg b.i.d for 3 days in 2 anisakiasis patients, i.e., mebendazole alone in 1 case and supplementary to surgery in another case. The results were satisfactory in both cases. Moschella et al. [93] used the same dosage of mebendazole for post-operative treatment of a patient with intestinal anisakiasis. Of considerable interest to note is that mebendazole was recently highlighted to be a more effective drug than albendazole and flubendazole in reducing lesion occurrence in experimentally infected rats [94].
Trichinosis
The efficacy of albendazole against Trichinella spiralis infection was first confirmed by McCracken [95] in experimental mice; treatment with 50 mg/kg for 5 consecutive days reduced the number of muscle larvae by 67% compared to controls. However, this efficacy of albendazole was worse than that of mebendazole (at the same dosage regimen) which revealed 96% reduction rate of muscle larvae [95]. The therapeutic usefulness of albendazole for human patients in combination with steroids was investigated during an outbreak of trichinosis in France by Fourestie et al. [96]. The regimen of albendazole was 400 mg daily for 3 days and then 800 mg daily for 15 days. Another study in France reported use of albendazole at the dose of 13±2.6 mg/kg daily for 8 days with satisfactory therapeutic effects with no side effects [97]. Thereafter, the recommended regimen of albendazole for treatment of human trichinosis has been settled at 800 mg (15 mg/kg) daily for 8–14 days [76,98] or 10–15 days [99,100] Albendazole is poorly absorbed through the intestinal tract especially in humans [3]. Thus, in order to increase the bioavailability of the drug in the plasma and enhance the therapeutic efficacy, trials have been made by co-administration with methimazole [101], using solid dispersion and cyclodextrin complexation techniques [102] or chitosan coated nanostructured lipid carriers [103].
Mebendazole at the dose of 50 mg/kg daily for 8–14 days was shown to be effective against migrating larvae of T. spiralis in experimental mice [104]. The clinical trial of mebendazole (1,000 mg daily for 14 days) for human trichinosis was reported in 1977, and no side effects were observed [105]. Use of prolonged (2 courses) and a higher dose of mebendazole was reported in a human patient unresponsive to steroid therapy [106]. In experimental mice, mebendazole showed better effects to kill muscle larvae than albendazole [95]. However, the effect of mebendazole was limited in human trichinosis because it was effective only on newborn larvae in lymph and blood vessels but not on encapsulating larvae in muscle cells [107]. The dosage of mebendazole for trichinosis has now been settled at 5 mg/kg daily for 10–15 days [99] or 200–400 mg t.i.d. for 3 days followed by 400–500 mg t.i.d. for 10 days [76,98,100].
Hepatic capillariasis
Albendazole and mebendazole were tried for treatment of hepatic capillariasis (frequently fatal especially in children), due to Capillaria hepatica, by observing reduction of egg deposition in the liver of experimental mice [108]. Both drugs were found to be effective, although mebendazole appeared to be more useful as the effects could be obtained using dosage regimens within the range recommended for humans; 10 times higher dosage was required for albendazole to obtain the same results [108]. However, clinical trial has been reported only in albendazole but not in mebendazole.
The first clinical use of albendazole (10–20 mg/kg daily for 20 days) was reported in a child case with only a limited success, which was followed by thiabendazole (25 mg/kg daily for 27 days) and prednisolone (10 mg daily) therapy with final survival of the patient [109]. Albendazole was again used in 2 patients; 400 mg daily for 21 days and 100 days (in combination with steroids) each, and the results were both satisfactory [110]. Thus, the drug of choice for hepatic capillariasis is currently thiabendazole or albendazole for 21 days [111]. The clinical use of mebendazole for human hepatic capillariasis remains to be tried.
Intestinal capillariasis
Human intestinal capillariasis due to Capillaria philippinensis has been treated with albendazole or mebendazole. In 1987, Cross and Basaca-Sevilla [112] used albendazole (400 mg daily for 10 days) in 16 intestinal capillariasis patients, and 15 were completely cured. One treatment failure case was retreated with mebendazole 400 mg daily for 20 days with success; however, they recommended albendazole as the drug of choice [112]. Thereafter, many workers used albendazole for treatment of this disease, including Lee et al. [113], Hong et al. [114], Bair et al. [115], Soukhathamvong et al. [116], Rana et al. [117], and Sadaow et al. [118]. The dosage regimen of albendazole has been 400–800 mg daily for 10–30 days.
The use of mebendazole for human intestinal capillariasis seems to be first tried by Alcantara et al. [119] in 1985. The patient experienced an obstinate course of the disease, including 16 episodes in 12 years, and was treated with thiabendazole, mebendazole, and flubendazole. The final treatment with mebendazole (200 mg daily) for a total of 8.5 months resulted in clinical and parasitological cures without further relapses [119]. Thereafter, mebendazole (200–400 mg daily for 20–30 days) was recommended as the drug of choice for intestinal capillariasis [115,120–122]. However, after albendazole was proved to be also effective, both drugs have been recommended as the drug of choice for human intestinal capillariasis by The Medical Letter [76].
Angiostrongyliasis
Use of anthelmintics in eosinophilic meningoencephalitis due to Angiostrongylus cantonensis infection (cerebral angiostrongyliasis in humans) has been controversial. Most patients have a self-limited course and recover completely, and analgesics, corticosteroids, and careful removal of cerebrospinal fluid at frequent intervals can relieve symptoms from increased intracranial pressure [76]. However, Hwang and Chen [123] suggested the usefulness of albendazole for treatment of A. cantonensis infection in children. Thereafter, a combination therapy of albendazole (200–1,000 mg daily for 7–14 days) with dexamethasone (or prednisolone) effectively treated eosinophilic meningoencephalitis patients [124–126]. Federspiel et al. [127] reviewed the literature of 22 A. cantonensis cases reported since 1988, and found that 12 of the 22 cases were treated with albendazole in combination with corticosteroids. Some studies [125,126] agreed that albendazole was useful for reducing the clinical symptoms, including headache. However, others do not recognize the superiority of using albendazole in combination with corticosteroids over corticosteroid treatment alone [127].
Mebendazole, combined with glucocorticosteroids, was reported to be effective in treatment of A. cantonensis infection; it appeared to shorten the course of infection though not the number of relapses [128]. Chotmongkol et al. [129] used mebendazole (10 mg/kg daily for 14 days) in combination with prednisolone in 41 patients, and headache completely disappeared in 37 (90.2%) patients within 14 days of treatment. However, Leone et al. [130] could not continue mebendazole treatment in a patient because of accentuation of the symptoms after the first dose of mebendazole; the patient was switched to steroid therapy. The usefulness of mebendazole for eosinophilic meningoencephalitis remains to be further defined.
In abdominal angiostrongyliasis due to infection with Angiostrongylus costaricensis, anthelmintics such as albendazole and mebendazole have not proven to be efficient, since the worms can cause lesions after their death inside the blood vessels [131].
Gnathostomiasis
Five species of Gnathostoma, including G. spinigerum, G. hispidum, G. doloresi, G. nipponicum, and G. binucleatum, have been reported to infect humans [132]. The first use of albendazole for treatment of human gnathostomiasis was reported in 1987 by Chitchang S. in a local journal (Thai) [133]. Subsequently, its usefulness (400–800 mg daily for 21 days) was confirmed with 93.9–94.1% CR in 100 gnathostomiasis patients in Thailand [133]. Similar results were reported either using albendazole alone (the same regimen) or ivermectin alone (0.2 mg/kg single or double doses) [134–137]. Chai et al. [138] used albendazole (1,200 mg daily for 21 days) in combination with ivermectin (0.2 mg/kg single doe) in 38 Korean patients who immigrated to Myanmar with satisfactory results. However, superiority of albendazole alone or ivermectin alone for treatment of human gnathostomiasis has been controversial [139,140]. However, albendazole stimulates the outward migration of the gnathostome larva, thus making it more accessible and possibly amenable to excision [141].
Mebendazole has seldom been used for treatment of human gnathostomiasis. However, in a recent report from Madagascar, a G. spinigerum 3rd-stage larva was surgically extracted from an eye of a woman, and then mebendazole (200 mg daily for 21 days) was prescribed to treat probable further infection by Gnathostoma parasites and also to cure a wide spectrum of intestinal nematode infections [142].
Gongylonemiasis
Human gongylonemiasis, due to Gongylonema pulchrum, occurs on the lips, gums, tongue, and palate eliciting the sensation of a moving worm (s) in the buccal cavity [143]. Removal of the parasite may be curative; however, because of possible additional worms, anthelmintic drugs, including albendazole (400 mg daily for 3 days), have been prescribed [143–147]. However, in a patient having esophageal symptoms, this albendazole regimen failed to prevent recurrence, and a prolonged administration of albendazole (400 mg daily for 30 days) was needed to obtain a complete cure [148].
In experimental rabbits, mebendazole (70 mg/kg daily for 3 days) showed 22.8% reduction of worm burdens of G. pulchrum; however, levamisole (8 mg/kg single) revealed a higher reduction of 63.2% [149]. Clinical trials for mebendazole in human patients have been unavailable.
Thelaziasis
Human thelaziasis is mostly due to infection with Thelazia callipaeda and less frequently by Thelazia californiensis [150]. Mechanical removal of worms directly from the eyes of humans or animals is the first treatment of choice [150]. Nonetheless, this does not guarantee complete extraction of all parasites from the eyes; therefore, anti-parasitic medication is sometimes necessary [150]. Moxidectin and milbemycin oxime are the drugs of choice for thelaziasis; but mebendazole (20 mg/kg daily for 3 days) was also shown to be effective for cure of the infection in dogs [151]. In a human patient complaining of foreign body sensation in the right eye, 3 T. callipaeda worms were retrieved, and albendazole (400 mg daily for 3 days) was prescribed for a supplementary purpose; this treatment was repeated 1 more time 2 weeks later [152]. However, the efficacy of albendazole and mebendazole in treating hidden T. callipaeda infection in the eyes remains to be further clarified.
Dracunculiasis
The treatment of dracunculiasis (due to Dracunculus medinensis) is largely dependent upon mechanical removal of worms (winding the emerging worm around a stick) from the subcutaneous tissue [153]. Medical treatment is usually not necessary, and drugs attacking the worm inside the host body may cause anaphylactic reaction due to the death of larvae [153]. However, dracunculiasis typically occurs in remote areas without easy access to medical attention, and anthelmintic drugs may be beneficial for such cases and also in mass, community-based control trials [154]. Nevertheless, albendazole, thiabendazole, metronidazole, ivermectin, and DEC showed no significant effects in managing this disease in animals, thus were poor candidates for clinical trials [153,154].
Cerebral and subcutaneous cysticercosis
Albendazole began to be used for treatment of human neurocysticercosis caused by the metacestode of Taenia solium in 1987 by Escobedo et al. [155]. They observed that albendazole (15 mg/kg daily for 30 days) was highly effective for treatment of patients with brain parenchymal cysticercosis; it was also effective in patients who had shown poor therapeutic response to praziquantel [155]. Sotelo et al. [156] recommended starting a course of albendazole (15 mg/kg daily for 30 days) therapy, and 3 months later, if parenchymal cysts are still detected, a course of praziquantel should be tried. Even a shorter course of treatment with albendazole (15 mg/kg daily for 3 days) was as effective as the 30-day course treatment [157]. Currently, the recommended regimen of albendazole for treatment of neurocysticercosis is 15 mg/kg daily for 8 days (7–15 days) in combination with steroids if necessary [22,158,159] (Table 3). Albendazole was also found to be effective for subarachnoid and ventricular cysticercosis [160]. The antiparasitic activity of albendazole is closely related to bioavailability of its active compound, albendazole sulfoxide; in this form, the drug passes through the blood-brain barrier and reaches the central nervous system where it exerts its therapeutic effects [161].
Albendazole (15 mg/kg daily for 30 days) was highly effective against dermal and cutaneous cysticercosis; most subcutaneous nodules disappeared or were markedly reduced in size [162]. Almost the same dose of albendazole (15 mg/kg daily for 28 days), in combination with steroids tapered over a period of 28 days, brought about complete recovery of ptosis due to extraocular muscle cysticercosis in the right eye [163]. However, it is of note that using corticosteroids in experimental cysticercosis by Taenia crassiceps negatively affected the efficacy of cysticidal activity of albendazole [164].
Mebendazole was assessed for use in treatment of cysticercosis in humans [165] and animals [166]. Some cures were documented but the results were equivocal and temporary inhibition of cyst growth or absence of a detectable effect were the more likely sequelae [165]. Thus, mebendazole alone is not recommended for human cysticercosis. However, mebendazole in combination with praziquantel has been proposed to be a promising alternative for treatment of human or animal cysticercosis [167].
Echinococcosis
The primary choice of treatment for human cystic echinococcosis (=cystic hydatidosis) due to Echinococcus granulosus infection is surgery unless the cyst is multiple or inoperable [168]. Chemotherapy with anthelmintics, including albendazole, mebendazole, praziquantel, or nitazoxanide, is an alternative to surgery and for a pre-operative or post-operative treatment, as well as management of inoperable alveolar echinococcosis due to Echinococcus multilocularis by long-term chemotherapy [168,169]. Albendazole has been used since 1983 [170,171]. A great number of case studies were performed, and valuable results were obtained [7,168,169,172–175]. The dosage regimen of albendazole in the first 2 pilot studies was 10–14 mg/kg daily for 2–39 days [170] and 10 mg/kg daily for 5–8 days [171]. Currently the recommended dose is 400 mg twice daily for 3–6 months with 14 days of break for cystic echinococcosis and 400 mg twice daily for minimum 2–3 years for alveolar echinococcosis [174]. In the meantime, combined albendazole and praziquantel treatment was tried with good results [176,177]. However, because of toxicity issues the use of this drug combination needs further research [175]. On the other hand, percutaneous drainage method was introduced as an alternative therapy for patients with hepatic hydatid cysts who cannot undergo surgery [175,178–182]. The procedure consists of puncture (P), aspiration (A), injection of scolicidal agent (I), and reaspiration (R), so-called PAIR, and this method (PAIR) in combination with albendazole or mebendazole has been evaluated as a highly useful strategy for managing cystic hydatidosis [174,175,181,182]. Now, 4 treatment options for human cystic echinococcosis have been proposed; 1) surgery, 2) PAIR, 3) chemotherapy with albendazole, mebendazole or other anthelmintics, and 4) watch-and-wait for inactive or silent cysts [169].
Mebendazole was first shown to be effective in killing hydatid cysts (E. granulosus) in experimental mice [183]. Human cystic echinococcosis began to be treated with mebendazole (400–600 mg t.i.d. for 21–30 days), and regression of the cysts and clinical improvement were observed [184]. However, it often remained speculative whether the regressed cysts were completely destroyed [185]. Higher dose (50–70 mg/kg daily) of mebendazole with longer duration (6–24 months) was tried in human cystic echinococcosis, and its efficacy was compared with that of albendazole (10 mg/kg daily for 5.5 months) [172]. The treatment was partially or completely successful in 16 (57.1%) of 28 patients treated with mebendazole and in 20 (87.0%) of 23 patients treated with albendazole [172]. Other studies also supported superiority of albendazole over mebendazole for medical treatment of human cystic hydatid disease [169]. However, in children, mebendazole (50 mg/kg daily for 9–18 months) was appropriate to use due to the lack of side effects, high therapeutic effects, and low risk of recurrence [186]. In addition, a combination of mebendazole (3,000 mg daily for 30 days) with praziquantel (1,800 mg daily for 15 days) with variable resting periods was suggested to be a good pharmacological alternative in the medical management of cystic echinococcosis [187]. The regimen of mebendazole in human cystic echinococcosis was studied by Vutova et al. [188]; it was concluded that the CR was increased both with dosage (from 30 to 70 mg/kg daily) and duration (from 6 to 24 months). PAIR plus pre- or post-operative mebendazole therapy brought about greater clinical and parasitological efficacy compared with surgery alone [168,181,182]. In alveolar echinococcosis, long-term mebendazole therapy (45–48 mg/kg daily for 13 years) was suggested to have a good effect [189].
LYMPHATIC FILARIASIS AND OTHER FILARIASIS
Lymphatic filariasis
Lymphatic filariasis is caused by infection with filarial nematodes, especially, Wuchereria bancrofti, Brugia malayi, and less commonly Brugia timori, transmitted by vector mosquitoes [190]. The use of anthelmintic drugs is to kill microfilariae in the blood of patients (asymptomatic or symptomatic) which is beneficial for reduction of disease transmission to other people. The conventional chemotherapy for this purpose was 2-week course of diethylcarbamazine (DEC) (6 mg/kg daily); however, single doses of DEC (4, 6 or 8 mg/kg) were also found to be as effective as in long-term reduction of microfilaria levels up to 1 year after treatment [191]. This (DEC 6 mg/kg in a single dose) had been for some time the mainstay of filariasis control programmes in the field [192]. In the meantime, 2-drug treatment with DEC and ivermectin both in single doses was shown to be more effective than treatment with either drug alone, yielding up to 99% clearance of microfilaremia up to 1 year after treatment [192,193]. However, significant concerns remained (severe side reactions, even fatal, occur due to DEC), and DEC made any multidrug regimen unsafe for community-wide use in Africa or elsewhere onchocerciasis or loiasis might coexist with bancroftian filariasis [192].
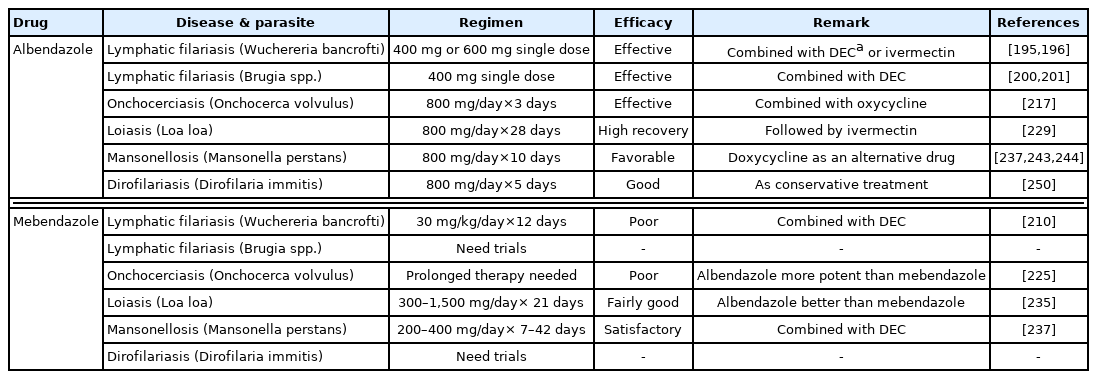
Jayakody et al. [194] was the first who formally conducted a study on the effectiveness of albendazole (800 mg daily for 21 days) in comparison with DEC (6 mg/kg daily for 21 days) in W. bancrofti infection in humans. Although the microfilaricidal activities of these drugs were dramatic, the majority of the patients experienced severe adverse effects such as acute pain, fever, and inflammation of the scrotal sac and adjacent tissue, presumably induced by dying adult worms and microfilariae [194]. Therefore, this high dose and long-term use of albendazole was discouraged, and a single reduced dose (400 or 600 mg), in combination with either ivermectin (200–400 μg/kg) or DEC (6 mg/kg), was tried; this new regimen proved to have both long-term effectiveness and safety in decreasing microfilaremia in W. bancrofti infection [195,196] (Table 4). However, albendazole should not be given as a single agent for lymphatic filariasis [197] except in areas co-endemic with Loa loa in Central Africa [198] or co-endemic with Onchocerca volvulus. The use of albendazole (400 mg in single dose) in combination with either ivermectin (150–200 μg/kg) or DEC (6 mg/kg) in single doses was found to be equally effective as DEC in combination with ivermectin against W. bancrofti infection in Africa or elsewhere in the world [192,199]. The combination of albendazole (400 mg in single dose) and DEC (6 mg/kg single dose) was found to be useful for treatment of also B. malayi infection in India [200] and B. timori infection in Indonesia [201] where there is no onchocerciasis or loiasis. Gyapong et al. [202] encouraged the use of albendazole in combination with ivermectin where onchocerciasis is co-endemic, and albendazole and DEC elsewhere. Nevertheless, it was suggested by Tisch et al. [203] that adding albendazole to DEC would provide less reduction in microfilarial prevalence and density than adding ivermectin to DEC. Addiss et al. [190], Critchley et al. [204], and Macfarlane et al. [205] were of opinion that not enough evidence had been gathered on the effectiveness of albendazole, alone or in combination with other drugs, for killing or interrupting transmission of filarial worms (adults and microfilariae) up to 12 months after treatment, and further well-controlled studies should be done. Although albendazole will not treat symptoms in people already affected by filariasis [205], it is of considerable importance to see that a significant proportion of children with W. bancrofti infection in India had their lymphatic pathology reversed when given the combination of albendazole and DEC annually or semiannually for 2 years [206]. In addition, a new therapeutic regimen, consisting of 3 drugs together (albendazole, DEC, and ivermectin), was tried for W. bancrofti filariasis, which was more effective in clearing microfilariae from the blood of the patients compared with a classical regimen, which consisted of albendazole and DEC, in Côte d’Ivoire [207] and India [208].
Wolbachia endosymbiotic bacteria is closely associated with worm development, embryogenesis, fertility, and viability of filarial nematodes and synergistically involved in the pathogenesis and clinical features in filariasis patients [209]. The use of doxycycline (200 mg daily for 21 days) to kill Wolbachia endosymbionts in combination with albendazole and ivermectin single doses showed effectiveness in inducing a long-term reduction of microfilaria levels in patients with W. bancrofti infection [209].
Mebendazole has seldom been used for treatment of lymphatic filariasis. Sarma et al. [210] used mebendazole (30 mg/kg daily for 12 days or weekly for 1 year) in combination with DEC but did not find any beneficial effect in most patients with clinical disease caused by W. bancrofti infection in India.
Onchocerciasis
The first drug of choice for onchocerciasis (Onchocerca volvulus infection) is ivermectin (150–200 μg/kg single dose) [211]. Albendazole (800 mg daily for 3 days) had been shown to be an alternative drug having a positive effect on prolonged suppression of skin microfilarial counts [212]. A prolonged course of albendazole treatment (400 mg daily for 10 days) in 26 patients caused changes in microfilarial densities over a year period, presumably by interfering with embryogenesis, though not directly killing microfilariae [213]. However, a combination of shorter duration of albendazole (800 mg daily for 3 days) with ivermectin (150 μg/kg single dose) given 1 week apart did not produce any additional effect when compared with ivermectin alone [214]. Another trial using albendazole (400 mg single dose) with ivermectin (200 μg/kg single dose) given concurrently also produced no advantage over ivermectin alone [211]. Repeated use (annually or semiannually) of albendazole (800 mg single dose) with ivermectin (200 μg/kg single dose) for 2 years also showed no better results than ivermectin alone for sterilizing and killing adult worms or achieving sustained microfilarial clearance [215]. In the meantime, doxycycline (100 mg daily for 35–42 days or 200 mg daily for 28 days) was shown to be effective in reducing the bacterial symbiont Wolbachia and finally in long-term (>24 months) sterilization of female O. volvulus worms and an absence of skin microfilariae [216]. A combination of doxycycline (200 mg daily for 21 days) with albendazole (800 mg daily for 3 days) produced better efficacy than either drug alone [217]. Fischer et al. [218] suggested a possibility that a new drug combination (ivermectin, DEC, and albendazole; IDA) which has been shown to be effective for control of lymphatic filariasis may also be effective for clearance and suppression of microfilariae of O. volvulus.
Mebendazole was found to be ineffective against O. volvulus in a Chimpanzee model; neither microfilariae nor adult worms on the skin were killed perhaps because the drug is poorly absorbed from the alimentary canal [219]. In human patients, mebendazole (200–300 mg daily for 6–30 days) alone or in combination with levamisole had no significant effects on the microfilariae or adult worms [220,221], although a high dose of mebendazole was suggested to be useful and relatively safe for therapy of onchocerciasis [222]. However, neither mebendazole alone nor its combination with levamisole was strongly macrofilaricidal in the skin [223]. Further development of mebendazole for chemotherapy of onchocerciasis was abandoned because of the length of therapy and the brief duration (3 months) of the effects on adult worms [224]. Albendazole is much more potent than mebendazole, its significant activity is achieved with a single dose, and prolonged therapy like mebendazole is unnecessary [225].
Loiasis
The filaricidal activity of albendazole (400 mg daily for 21 days) in human loiasis (Loa loa) was first confirmed from a double-blind, placebo-controlled clinical trial in 1993 in Benin [226]. In addition, albendazole (400 mg daily for 21 days) was found to be useful for loiasis refractory to DEC treatment [227]. Albendazole is considered to be embryotoxic resulting in a slow decrease of microfilaremia [226]. Arrey-Agbor et al. [228] reported a case of spontaneous encephalopathy due to loiasis whose microfilaremia was dramatically reduced with 21-day course of albendazole therapy. Gobbi et al. [229] obtained a high rate of recovery among 16 imported loiasis cases in Italy using a 28-day course of albendazole (800 mg daily) treatment followed by ivermectin (200 μg/kg in single or multiple doses). However, a 21-day course of treatment is not suited for population-based chemotherapy [230]. A reduced course of treatment with albendazole (400 mg daily for 3 days) was tried in 99 infected people in Cameroon but the results were not so satisfactory [230]. A 3-day course regimen with a higher dose (800 mg daily) given in 47 patients in Cameroon also revealed no significant change in the overall microfilarial loads [231].
The first trial of mebendazole (2 g daily for 21–28 days) for treatment of loiasis was done by Richard-Lenoble et al. [232] in 1985 which reported a slight decrease in microfilaremia, although the same mebendazole regimen (2 g daily for 21 days) revealed no significant decrease in microfilaremia [233]. Mebendazole did not show any difference in efficacy according to different dosage, from 300 mg to 1,500 mg daily, if used for 21 days, and 16 (84.2%) of 19 enrolled patients revealed complete or partial reduction of microfilaremia [234]. The microfilarial reduction was more pronounced in patients treated with albendazole (600 mg single dose) than those with mebendazole (100 mg b.i.d. for 1 day) [235]. The 21-day course of mebendazole treatment may constitute a preliminary treatment to progressively decrease the microfilarial density below the threshold which ivermectin or DEC can be safely administered [236].
Mansonellosis
Three species of Mansonella, including M. perstans, M. streptocerca, and M. ozzardi, are responsible for causing human mansonellosis [237]. Among them, M. perstans is the most important and most difficult species to treat [237] and relatively resistant to standard antifilarial agents, including DEC, ivermectin, albendazole, and mebendazole [238]. Albendazole (800 mg daily for 3–10 days) was first tried for M. perstans filariasis in 1992 but the results were not so satisfactory [239]. However, 2 long courses of albendazole (800 mg daily for 45 days) with 14 days’ rest were effective in a patient with M. perstans filariasis [240]. The same dose of albendazole given for 10 days produced favorable results in 47 patients with decreased microfilaremia until 90 days after treatment [241]. However, a single dose of albendazole (400 mg) and ivermectin (150–200 μg/kg) combined and each drug alone revealed low efficacies [242]. To treat the endosymbiotic Wolbachia bacteria, doxycycline began to be used as a promising alternative drug since the 2010s, alone or in combination with anthelmintics [237,243,244].
Mebendazole (200 mg daily for 42 days) was first shown to markedly reduce the number of microfilariae of M. perstans, and a combined use of mebendazole (200–400 mg daily for 7 days) with levamisole cleared the blood microfilaremia within 7 days [220]. Nine patients with M. perstans infection were also successfully treated with mebendazole (200 mg daily for 28–42 days) [245,246]. In another study, all of 16 cases infected with M. perstans revealed dramatically reduced parasitemia after mebendazole (300–1,500 mg daily for 21–45 days) treatment [234]. The efficacy of different anthelmintics against M. perstans filariasis was evaluated in 165 patients, and mebendazole (200 mg daily for 28 days) appeared to be more active than DEC (400 mg daily for 21 days) in eliminating the disease; a combined use of mebendazole (100–200 mg daily for 14–21 days) with DEC (400 mg daily for 21 days) produced better effects than mebendazole or DEC alone, with no increase of side effects [247]. Thus, mebendazole alone [238] or in combination with DEC is currently acknowledged as the most effective antiparasitic treatment to treat M. perstans infection [237].
Dirofilariasis
In human dirofilariasis (due either to Dirofilaria immitis or Dirofilaria repens), anthelmintic treatment is generally not recommended, and surgery is recommended if needed and feasible [248,249]. Antibacterial drugs, including doxycycline, may be useful to treat Wolbachia symbionts [248]. Surgery was not possible in 24 of 266 dirofilariasis patients in Russia because of active migration of parasites in tissues, and in such cases albendazole (800 mg daily for 5 days) in combination with doxycycline (200 mg daily for 5 days) were prescribed for conservative treatment in 22 of the 24 patients with good results [250]. Surgery was done in 4 cases of pulmonary dirofilariasis, and post-operative medication was given with albendazole alone or in combination with DEC and doxycycline [251]. Mebendazole has seldom been tried for human dirofilariasis.
TREMATODE INFECTIONS
Fascioliasis
The treatment of choice for human fascioliasis (due to either Fasciola hepatica or Fasciola gigantica) currently is triclabendazole [15,252,253]. Praziquantel revealed no efficacy at all or only low-to-moderate CRs [252]. The efficacy of albendazole was first documented in the 1970s-1980s against fascioliasis in animals, including cattle and sheep [1,254,255]. Subsequently, albendazole began to be used for human fascioliasis; however, most of the treatments were unsuccessful [256–258]. Up to the present, according to available literature, only 2 human infections were successfully treated with albendazole (1,200 mg daily for 7 days) [17,259] (Table 5). Meanwhile, triclabendazole resistance of F. hepatica in livestock and patients was reported mainly from Western Europe [260], and use of albendazole was encouraged in such cases [261].
Mebendazole was rarely tried for treatment of human fascioliasis [15]. In 1 patient, a very high dose of mebendazole (4 g daily for 21 days) could treat human fascioliasis successfully with prompt normalization of eosinophil counts, liver enzymes abnormalities, and specific serologic test, together with disappearance of liver necrosis [20].
Clonorchiasis and opisthorchiasis
The drug of choice for clonorchiasis (due to Clonorchis sinensis) and opisthorchiasis (due to Opisthorchis viverrini or Opisthorchis felineus) is praziquantel [10,15,69]. Albendazole and mebendazole have also been used for these liver fluke infections [10,15]. In C. sinensis infection, a single dose of albendazole (400 mg) was almost ineffective, but multiple doses (for example, 400 mg t.i.d. for 3 days or 600 mg t.i.d. for 5 days) showed 84.6% and 100% CR and 99.9% and 100% ERR, respectively [13]. Similar results were reported; 5 mg/kg or 10 mg/kg b.i.d. for 7 days brought about 90% and 100% CR, respectively [262]. In O. viverrini infection, albendazole (400 mg b.i.d. for 3 or 7 days) revealed fairly high CR (60% or 96%) and ERR (91% or 92%) at day 14 post-treatment, respectively, when examined by Stoll’s method [14]. Therefore, the regimen of albendazole for treatment of clonorchiasis and opisthorchiasis should be 800–1,200 mg daily for 5–7 days, and it is not so feasible for MDA.
Mebendazole was tried for treatment of clonorchiasis and opisthorchiasis. A single dose of mebendazole (100 mg) was ineffective against C. sinensis infection [18]. Higher doses of mebendazole (30 mg/kg daily for 21–28 days) were found to be highly effective against O. viverrini infection with 96.3% CR [263]. However, this long-term medication is not useful for mass chemotherapy.
Intestinal fluke infections
For treatment of human intestinal fluke infections, including heterophyidiasis, echinostomiasis, neodiplostomiasis, and gymnophalloidiasis, praziquantel (10 mg/kg single dose) is the drug of choice [69], and the efficacy is usually satisfactory. However, albendazole was also tried for treatment of heterophyid fluke infections, including metagonimiasis due to Metagonimus yokogawai [13]. Its therapeutic efficacy was not so satisfactory with a single dose of 400 mg with 33.3% CR and 60.0% ERR; however, a 2-day regimen with daily dose of 400 mg revealed CR of 61.1% and ERR of 89.2%, respectively [13]. Albendazole showed similar effects on other heterophyid fluke infections. For example, Haplorchis taichui and Haplorchis yokogawai adult specimens were collected from the diarrheal stool of patients after treatment with albendazole (400 mg daily for 1 or 3 days) [264]. Adult echinostomes, including Echinostoma ilocanum and Artyfechinostomum malayanum, were also recovered from the diarrheal stool of 3 patients treated with albendazole (800 mg daily for 3 or 7 days) [14].
A long-term use of mebendazole (400 mg daily for 20 days) was also found to be effective for treatment of echinostome (E. ilocanum) infections in humans and animals [265].
PROTOZOAN INFECTIONS
Giardiasis
In vitro growth of Giardia spp. (G. lamblia, G. duodenalis, or G. intestinalis) was found to be highly sensitive to albendazole and mebendazole which had 30- to 50-fold higher activity than metronidazole, quinacrine, or tinidazole [266,267]. It was also shown that albendazole is active against G. duodenalis in vivo of mice [268] and dogs [269]. Albendazole seemed to damage the cytoskeletal elements of the ventral disk (=sucking disk) of G. duodenalis leading to loss of its ability to adhere to the intestinal villi and obtain nutrients [270]. A preliminary study on clinical use of albendazole (200–500 mg daily for 3 days for adults and 100 mg daily for 3 days for children) for giardiasis patients revealed promising results [271]. Moreover, in children infected with G. duodenalis, treatment with a single dose of albendazole (600–800 mg) revealed 62.1–74.6% CR, whereas multiple doses of albendazole (400 mg daily for 3 or 5 days) produced higher effects, 81.0–94.8% CR [272] (Table 5). Currently, the recommended dosage of albendazole for giardiasis is 400 mg daily for 5 days for adults or 10 mg/kg daily for 5 days for children [273]. Synergistic effects were obtained by a combined use of albendazole and metronidazole [274,275] and albendazole and secnidazole in treatment of patients who failed conventional treatments [276]. However, using suboptimal doses of albendazole may account for an increase in the prevalence of giardiasis in some areas and development of drug resistance [277].
The therapeutic effect (40% CR) of mebendazole (200 mg daily for 3 days) against human G. lamblia infection was first reported by Hutchison et al. [278] in 1975. Later, a higher dose of mebendazole (600 mg daily for 3 or 5 days) was used, and 38 of 40 patients (26 were below 12 years old) were cured (95% in CR) [279]. Mebendazole (600 mg daily for 5 days) was not effective for treatment of giardiasis in adult patients [280]. The efficacy of mebendazole was reported in another study; 21 (91.3%) of 23 patients given mebendazole (600 mg daily for 5 days) were cured, whereas 18 (85.7%) of 21 patients given metronidazole (600 mg daily for 5 days) became free from infection [281]. It seemed that a higher dose of mebendazole may be needed in adult patients [281].
Vaginal trichomoniasis
The ultrastructural changes of Trichomonas vaginalis trophozoites after treatment with albendazole were studied by Oxberry et al. [270] in 1994 in comparison with those that occurred in G. duodenalis trophozoites. In G. duodenalis trophozoites, albendazole seemed to damage the cytoskeletal elements of the ventral disk; however, in T. vaginalis trophozoites, such a ventral disk is lacking and the action of albendazole did not seem to be as great as that [270]. Another in vitro study revealed higher efficacy of mebendazole in killing T. vaginalis trophozoites than albendazole [282]. However, it is of note that albendazole and coenzyme B12 have been suggested to be helpful for treatment of patients with highly metronidazole (or tinidazole)-resistant T. vaginalis infections [283].
Antimicrotubular drugs, including mebendazole, flubendazole, and thiabendazole, were tested in vitro on their effects on growth and viability of T. vaginalis in 1985 [284]. Mebendazole was shown to be active at a lower concentration than albendazole [282]. Mebendazole was also shown to be effective against metronidazole-resistant and metronidazole-sensitive T. vaginalis strains in vitro [30,285]. However, in clinical use, mebendazole failed to cure 2 T. vaginalis patients who did not respond to metronidazole treatment [286].
Cryptosporidiosis
Human cryptosporidiosis is mainly caused by Cryptosporidium parvum and Cryptosporidium hominis. The albendazole and mebendazole susceptibility appeared to be low in C. parvum, whereas it was high in Encephalitozoon spp. and G. duodenalis [282,287]. It was demonstrated that the number of C. parvum parasites in histologic sections of intestine from mice treated with albendazole was significantly lower than that in untreated control mice; however, albendazole was not efficacious in clearing C. parvum infection in mice [288].
Microsporidiosis
Human microsporidiosis is caused by variable species of more than 9 genera belonging to the phylum Microsporidia (resembling fungi); the most important genera include Encephalitozoon, Enterocytozoon, Nosema/Vittaforma, Pleistophora, Anncalia (syn. Brachiola), and Microsporidium [289]. The therapeutic efficacy of albendazole (800 mg daily for 28 days) against human microsporidiosis due to Enterocytozoon bieneusi was first suggested by Blanshard et al. [290] in 1992. Nacey et al. [291] reported favorable results of albendazole (800 mg daily for 30 days) in a case of naso-ocular microsporidiosis due to Encephalitozoon cuniculi or Encephalitozoon hellem. The albendazole efficacy against human E. cuniculi infection was repeatedly confirmed [292,293]. The albendazole susceptibility of E. cuniculi and E. hellem was predicted by the presence of 2 β-tubulins, Glu-198 and Phe-200 [282]. Notably, however, E. cuniculi genotype III showed albendazole resistance in experimental mice [27]. Intestinal infection with Encephalitozoon intestinalis (syn. Septata intestinalis) also responded well to albendazole (800 mg daily for 28 days) [294,295], although in some patients only transient effects were seen [26]. A shorter duration treatment of albendazole (800 mg daily for 21 days) was also effective in treatment of E. intestinalis infections in patients with AIDS and delayed the occurrence of relapse, and a few relapsed cases could be cleared from infection using a second course of albendazole for 21 days [296]. Similar results were reported by Leder et al. [297]. Further shorter duration of albendazole treatment (15 mg/kg daily for 7 days) was tried and found to be effective in treatment of Microsporidium diarrheal patients [298]. However, E. bieneusi infection was not successfully cured using albendazole (800 mg daily for 28 days), with parasites not completely cleared in small-bowel biopsies of the patients [299–302]. The drug of choice for E. bieneusi infection is fumagillin or its analog TNP-470 [289,303, 304]; however, fumagillin may cause significant bone marrow toxicity [305]. A combination of albendazole and fumagillin could successfully treat a case of disseminated Anncaliia algerae microsporidial infection [306].
Mebendazole was tested for its efficacy on a microsporidian species, E. intesinalis, cultured in vitro using Vero cells; it was highly active in killing the spores of E. intestinalis [307]. However, mebendazole is poorly absorbed and thus it is unclear whether this drug would provide a suitable alternative to albendazole [307].
TOXICITY OF DRUGS
Albendazole
Very few adverse events have been reported by treatment with albendazole [53]. At the recommended doses used for intestinal helminths, all reported events were mild and self-limiting (epigastric pain, diarrhea, headache, nausea, abdominal pain, dizziness, vomiting, lethargy, constipation, leucopenia, pruritus, etc.), and none have been serious or life-threatening [53]. Most problems are associated with higher dose treatment for systemic infections where problems of drug-parasite interactions cause specific syndromes such as abnormal liver functions (for example, in echinococcosis), central nervous system symptoms (in neurocysticercosis), or hematologic side effects [53,308–312]. Fatalities are extremely rare, and have almost entirely been associated with severe underlying diseases or AIDS-related infections [53]. Importantly, however, a pilot study using albendazole (10 mg/kg daily for 28 days) for treatment of 7 colorectal or hepatocellular carcinoma patients reported fatality in 1 patient presumably due to albendazole toxicity, in particular, severe neutropenia [37]. Further, the maximum tolerated daily dose of albendazole was found to be 2,400 mg (for 14 days in a 21-day cycle) in 6 patients with advanced cancers, and myelosuppression, including neutropenia, leukopenia, lymphopenia, and thrombocytopenia, was the main dose limting toxicity in 2 of the 6 patients [313].
Notably, however, even a single dose treatment (for empirical or seasonal use) of albendazole (400 mg) could cause acute liver toxicity in adult patients [39,314–318] as well as in children [319–322]. Such liver toxicity may be recurrent each time after administration of albendazole [317,318,320,321]. In another rare instance, acute renal failure occurred after taking albendazole (800 mg daily for 2 days) to treat trichinosis [323]. Albendazole is known to be a teratogen and fetal toxicant in experimental animals at very high doses [3]. Albendazole is contraindicated during pregnancy in humans [3,11].
Mebendazole
Mebendazole is a highly safe anthelmintic drug if used in recommended doses. One of the most important adverse effects is hepatotoxicity, although its incidence is very rare [324]. It was reported in treatment of echinococcosis for which long-term mebendazole therapy was needed [36,325,326]. Hepatotoxicity was also reported in a patient with Gilbert’s syndrome (inherited disorder of bilirubin glucuronidation) who was prescribed with mebendazole (200 mg daily for 3 days) to treat pinworm infection [324]. In this patient, a diminished hepatic glucuronidation of mebendazole caused the increase in the level of unconjugated toxic metabolites and the consequent induction of the liver damage [324]. However, in children patients with hydatid disease, even long-term mebendazole treatment (50 mg/kg daily for 9–18 months) was safe without significant side effects [186]. Granulocytopenia, alopecia, pruritus, skin abscess, and arthritis were reported in patients treated with high doses of mebendazole [36,238,327]. Importantly, a combined use of mebendazole (>500 mg) and metronidazole (>500 mg) is prohibited because severe and rare fatal adverse events such as Stevens-Johnson syndrome (or toxic epidermal necrosis) may occur [328]. The risk increased with increasing doses of metronidazole but not mebendazole, and there may be a synergistic interaction between mebendazole and metronidazole [328]. In experimental rats, mebendazole has been shown to be a teratogen if given very high doses although not in rabbits [3]. In human patients, mebendazole is contraindicated during pregnancy [3,34].
DRUG RESISTANCE
Hookworms of veterinary importance
Large scale use, use of inappropriate doses, and a failure to alternate treatment with other drug classes have caused selection for resistant parasite populations, which render previously effective doses of anthelmintic drugs ineffective [329,330]. This kind of drug resistance has been reported in benzimidazoles, including albendazole and mebendazole. Earlier studies on the drug resistance to benzimidazoles were performed on intestinal nematodes of veterinary importance, in particular, Haemonchus contortus; the drug resistance in this nematode was found to be correlated with a single nucleotide polymorphism at codons 167, 198, or 200 of the β-tubulin isotype 1 gene [331,332]. This single nucleotide polymorphism in the β-tubulin isotype 1 gene was also found in Ancylostoma caninum [331]. However, a different resistance mechanism, although not yet elucidated, may be present in the drug resistance of intestinal nematodes, for example, in Ancylostoma ceylanicum [333]. The resistance might also arise in various ways, including point mutations or deletion of β-tubulin isotype 1 or 2, microtubule-associated proteins, or others [334,335].
Human-infecting hookworms
Regarding human-infecting intestinal nematodes, such as hookworms, the emergence of drug resistance has been suggested by various workers, including Bennett and Guyatt [336]. In treatment of human hookworm (Necator americanus) infections in Mali, mebendazole (500 mg single dose) gave disappointing results (22.9% in CR) presumably due to a drug resistance [337]. Thereafter, low efficacies of mebendazole or albendazole against hookworms were repeatedly reported; for example, in Pemba Island, Zanzibar [335,337], Vietnam [338], and Ghana [339]. In Pemba Island, the efficacy of mebendazole (500 mg single dose) against hookworms in schoolchildren had fallen over a period of 5 years, from 22.4% to 7.6% in CR and 82.4% to 52.1% in ERR, during which time the children were regularly treated [337]. However, molecular studies revealed no evidence for the β-tubulin mutation at amino acid residue 200 (Phe/Tyr) [335]. In Vietnam, a single (500 mg) or multiple doses of mebendazole (500 mg daily for 3 days) was found to have disappointing efficacy against hookworm infections in children (CR; 38% in mebendazole single dose group vs 33% in placebo group) and adults (CR; 26% in mebendazole 3-day group vs 35% in placebo group) [338]. Drug resistance was also shown in albendazole in adults; CR was 45% in single dose albendazole (400 mg) group and 79% in albendazole 3-day group in comparison with 35% placebo group [338]. In Ghana, poor efficacy of albendazole (400 mg single dose) was obtained in treatment of hookworms, 61.1% CR and 81.5% ERR, presumably due to drug resistance [339]. In N. americanus, isothermal diagnostic assays were developed to target single nucleotide polymorphisms at codons 167, 198, or 200 of the β-tubulin isotype 1 gene, to study on the drug resistance [41].
Trichuris trichiura
As to T. trichiura infection, Bennett and Guyatt [336] reviewed literature on the efficacy of albendazole and mebendazole and found that the CR of albendazole (400 mg single dose) was markedly lower in Asia than in Africa, Central and South America (33.3% vs 61.0%, respectively) probably due to a drug resistance. The low efficacy of mebendazole against T. trichiura was again reported in schoolchildren on Pemba Island; mebendazole (500 mg single dose) gave only 22.9% CR and 81.0% ERR [337]. Diawara et al. [340] confirmed different alleles at codon 200 in the β-tubulin gene (only 1 isotype has been found at this gene in T. trichiura) from T. trichiura worms (Kenya) and eggs (Panama) obtained from children. It was suggested that the gene mutation at codon 200 might be related with the drug resistance [340,341]. Also, a single dose of albendazole (400 mg) revealed highly variable efficacies against T. trichiura infection according to different countries, including Cameroon, Ethiopia, India, Tanzania, and Vietnam, with 21.0–88.9% CR and 39.2–93.1% ERR, partly due to drug resistance [342]. The drug resistance of T. trichiura to benzimidazoles was also suggested by Moser et al. [42,43]. They demonstrated that the CR and ERR of T. trichiura by albendazole (400 mg single dose) were 38.6% and 72.6% in 1995 which decreased to 16.4% and 43.4% in 2015, respectively [43]. In case of mebendazole, its efficacy (500 mg single dose) for T. trichiura infection was 42.1–44.4% in CR and 66.0–80.7% in ERR [42]. For a short-term control (4 months) of trichuriasis in schoolchildren, Myanmar, a single dose of albendazole (400 mg) appeared to be not enough, and a long-term MDA (>10 years), increasing albendazole dosage-regimen into 3-day course, or using an alternative drug or drug combination was recommended [46].
Ascaris lumbricoides
A reduced efficacy of albendazole (400 mg single dose) was also reported in A. lumbricoides in Rwanda for which parasite no drug resistance had been previously recognized [40]. The CR of A. lumbricoides infection in 1 of 12 schools was as low as 35.3%, and the average CR and ERR in 12 schools was 69.9% and 75.4%, respectively; however, no gene mutation was found at codons 167, 198, and 200 from 4 Ascaris β-tubulin isotype genes [41]. Reversely, Diawara et al. [341] identified a single nucleotide polymorphism at position 167 of A. lumbricoides β-tubulin.
Wuchereria bancrofti
Benzimidazole resistance-associated mutations were also found in the filarial nematode W. bancrofti; some of the microfilariae obtained from albendazole-treated patients revealed mutation at the position 200 of β-tubulin genotype [343].
Giardia sp
In Giardia sp., albendazole resistance was reported which was correlated with cytoskeletal changes but not dependent on mutation at codon 200 in β-tubulin gene [344].
ANTI-CANCER EFFECTS
Albendazole
The use of mammalian cell tubulin as a screen for potential anti-cancer drugs has been well documented [345]. Benzimidazole carbamates were shown to act against L1210 mouse leukemia cells in vitro via inhibition of tubulin polymerization [345]. Albendazole, one of the benzimidazoles, has the activity against parasite as well as mammalian tubulins and was shown to suppress in vitro and in vivo (mice) proliferation of hepatocellular carcinoma cells through inhibiting tubulin polymerization [38,346]. Ovarian [347] and prostate and colorectal cancer cells [348] were also suppressed by treatment with albendazole alone or albendazole plus 2-methoxyestradiol by inhibiting tubulin polymerization. Breast cancer and melanoma cell lines were susceptible to cytotoxic effects of albendazole, and a combined use of albendazole with other antitumor drugs may lead to an effective anti-cancer therapy [349,350]. In addition, albendazole was shown to act in vitro as a potent radiosensitizer in metastatic melanoma and small cell lung cancer cell lines [351] and had anti-angiogenesis effects on the cornea [352]. In Ehrlich carcinoma model in mice, albendazole induced oxidative stress promoting DNA fragmentation, triggering apoptosis, and inducing cancer cell death [33]. Further, albendazole exerted its anti-cancer activity in gastric cancer cell lines by disrupting microtubule formation and function to cause mitotic arrest and inducing cancer cell apoptosis [353] or by affecting STAT3 and STAT5 activation by pleiotropic mechanisms [354]. However, the anti-tumor activity of albendazole was cancer cell-type dependent; 13 of 14 human papillomavirus-negative head and neck squamous cell cancer cell lines responded well to albendazole, whereas only 3 of 6 human papillomavirus-positive head and neck squamous cell cancer cell lines responded to albendazole [355]. Albendazole also showed anti-tumor effects on cutaneous squamous cell carcinoma cells via regulation of endoplasmic reticulum stress and cancer stemness [356].
Compared with many experimental in vitro and in vivo (mice) studies, clinical application of albendazole in treatment of cancer patients has been limited. A pilot clinical study was performed in 9 patients with advanced colorectal carcinoma with liver metastasis (n=7), hepatocellular carcinoma (n=1), or mesothelioma/carcinoid tumor in the liver and pleura (n=1) [37] (Table 6). Out of 7 patients in whom serum tumor markers could be measured, albendazole treatment (10 mg/kg daily for 28 days) resulted in a decrease in carcinoembryonic antigen (CEA) or α-feto protein (AFP) levels in 2 patients, and these markers were stabilized in 3 patients [37]. In the remaining 2 patients, CEA or AFP levels, after initial stabilization during 5–10 days, began to increase again [37]. Long-term (28 days) treatment with albendazole was well tolerated, and there were no significant changes in hematological, kidney or liver function tests [37]. However, 3 of the 9 patients treated with albendazole developed severe neutropenia which was probably contributory to the death of 1 patient [37]. Another clinical study reported was a phase 1 clinical trial involving 36 solid (colorectal, gastric, mesothelial, or other) tumor patients refractory to conventional anti-cancer therapies, and different dosage regimens of albendazole (800 mg daily for 7–14 days, 1,600 mg daily for 14 days, 2,000 mg daily for 14 days, or 2,400 mg daily for 14 days) were tried [313]. The maximum tolerated dose of oral albendazole found in these patients was 2,400 mg per day (1,200 mg b.i.d.), and the recommended dose for further study was 2,400 mg daily for 14 days in a 21-day cycle [313]. However, 3 of 18 patients given 2,000–2,400 mg daily for 14 days developed neutropenia and other myelosuppression signs [313]. There is an on-going clinical trial for use of albendazole and mebendazole in Mexico (July 2011 to December 2023) for 250 patients who are considered untreatable, progressive, and fatal [35].
Myelosuppression, including neutropenia, leukopenia, lymphopenia, thrombocytopenia, and anemia, were seen in patients given albendazole at the daily dose of 2,400 mg which were found to be the main dose limiting toxicity [313]. Because of this toxicity limitation, mebendazole rather than albendazole has been more popularly recommended as a benzimidazole anti-cancer agent [34]. In addition, fenbendazole, a popular anthelmintic used in dogs and other animal species, has been proposed as a new potential microtubule interfering agent that displays anti-neoplastic activity in humans [357]. Large-scale clinical evidence is lacking but it has been mentioned that a human lung cancer survivor used fenbendazole for treatment of late-stage small cell lung cancer; however, the safety of fenbendazole for human use needs to be clarified [35].
Low solubility of albendazole in water is one of the main obstacles for use as an anti-parasitic as well as anti-cancer drug. Among various trials to overcome this problem, the most successful approach is to formulate or conjugate it into nanostructures or nanoformulations, in practice of improving aqueous solubility [358]. Albendazole-cyclodextrin complex, M1·albendazole, albumin nanoparticles, chitosan nanoparticles, solid lipid nanoparticles, PLGA nanoparticles, albendazole-copper oxide nanoparticles, and solid polymeric nanoparticles are examples of so far designed nanoformulations of albendazole [358,359].
Mebendazole
The anti-tumor activity of mebendazole was first tested by Mukhopadhyay et al. [360] by in vitro and in vivo (mice) experiments using human lung cancer cell lines. Mebendazole was a potent inhibitor of tumor cell growth with little toxicity to normal fibroblasts (W138) and human umbilical vein endothelial cells [360]. At the same time, Sasaki et al. [361] found that mebendazole actively inhibited microtubule assembly and spindle formation of human non-small cell lung cancer cells which led to mitotic arrest and death of cancer cells. Anti-tumor activity of mebendazole also seemed to be due to a decrease in angiogenesis of cancers [361]. The anti-tumor activities of mebendazole were also demonstrated in human adrenocortical carcinoma cells implanted into nude mice [362] and chemoresistant human melanoma cells [363]. Mebendazole was suggested to have advantages in a preclinical study of brain glioma-mouse model; mebendazole but not albendazole was able to significantly increase survival of mice having glioblastoma multiforme [364]. Five colon cancer cell lines and 3 non-malignant intestinal epithelial cell lines were tested for response to mebendazole, and selective anti-cancer activity was found [365]. The mebendazole activity against brain tumor cells (glioblastoma cell lines) was proven to be caused by inhibiting microtubule functions of the cells [366,367], and the survival of tumor-cell recipient mice was significantly increased by mebendazole treatment [366]. Mebendazole was also shown to be useful as a drug candidate for ovarian cancer therapy [368] as well as to synergize with docetaxel for prostate cancer treatment [369]. Collectively, mebendazole has been shown to not only exhibit direct cytotoxic activity, but also synergize with ionizing radiation and different chemotherapeutic agents to stimulate anti-tumoral immune responses which result in reduction or complete arrest of tumor growth, marked decrease of metastatic spread, and improvement of survival [34].
Many clinical trials with mebendazole are currently on-going [35]; however, published documents in treatment of cancer patients have so far been limited only to 2 case reports [370,371] (Table 6). The first report was published by Dobrosotskaya et al. [370] in the United States. The patient was a 48-year-old man with adrenocortical carcinoma with liver metastasis, and conventional treatments with mitotane, 5-fluorouracil, streptozotocin, bevacizumab, and radiation therapy failed and were intolerable [370]. Mebendazole (200 mg daily for 19 months) brought about regression of hepatic metastatic lesions with subsequent stability for 19 months, although the disease progressed after 24 months; however, the patient had no clinically adverse effects, and the quality of life was satisfactory [370]. A combined use of mebendazole with other cytotoxic drugs was suggested to have synergistic effects [370]. The second case was a colon cancer patient with liver and lung metastases; the patient was treated conventionally with capecitabine, oxaliplatin, and becacizumab but failed largely due to oxaliplatin-induced neuropathy [371]. Mebendazole therapy (200 mg daily) was continued for 42 days, and there were near complete remission of the metastases in the lungs and lymph nodes and a good partial remission in the liver [371]. Because of elevation of liver enzymes, mebendazole was stopped temporarily and then reintroduced at half dose; liver enzymes slowly decreased and the patient reported no adverse effects from mebendazole [371]. For future dosing and efficacies of mebendazole, at least 5 clinical trials are on-going in several countries, including 2 phase 1 clinical trials using mebendazole (500 mg chewable tablets, 3 times daily) on 24 and 21 high-grade glioma patients by The Johns Hopkins Hospital (NCT02644291, April 2013 to September 2025) and by The Johns Hopkins All Children’s Hospital, USA (NCT 0244291, May 2016 to June 2022), respectively, 1 phase 1/2 trial on 36 low- and high-grade glioma patients using mebendazole (50–200 mg/kg for 70 and 48 weeks, respectively) by Cohen Childrens’ Medical Center of New York, USA (NCT01837862, October 2013 to April 2020), 1 phase 2 trial on 250 patients with variable malignant diseases using mebendazole, albendazole or other drugs by Dr. Frank Arguello Cancer Clinic, Mexico (NCT02366884, July 2011 to December 2023), and 2 phase 3 trials using mebendazole as adjuvant treatment for 40 patients with stage 4 colorectal cancer in Egypt (NCT03925662, April 2019 to December 2028) and using mebendazole (100 mg daily for undefined duration) for treatment of 207 cancer patients by Care Oncology Clinic, UK (NCT02201381, April 2017 to May 2022) [35]. These trials are expected to bring about fruitful results on usefulness of mebendazole as a repurposed anti-cancer drug.
CONCLUSIONS AND PERSPECTIVES
Albendazole and mebendazole are highly useful broad-spectrum anti-parasitic (anti-fungal) as well as anti-cancer agents. These drugs have been widely used for treatment of human intestinal helminthiases (nematodes and cestodes), tissue helminthiases (nematodes and cestodes), filarial nematode infections, liver and intestinal trematode infections, and some kinds of protozoan infections. Recently, these drugs are highlighted because of their anti-cancer activities in vitro and in vivo of animals, and a few clinical anti-cancer trials have been documented. Further trials should be performed for their use as anti-cancer agents. Albendazole and mebendazole are highly safe if used in recommended doses as anti-parasitic agents. However, attention should be paid because hepatic toxicity may rarely occur in patients administered with only a single dose of albendazole. Increasing reports of drug resistance in parasites against albendazole and mebendazole are new emerging issues.
ACKNOWLEDGMENT
We are grateful to the staff of Institute of Parasitic Diseases, Korea Association of Health Promotion, Seoul, Korea who helped this study. We dedicate this review article to late Prof. Emer. Han-Jong Rim, Korea University, Seoul, Korea, a pioneer of anthelmintics and parasitology research, who recently passed away.
Notes
We have no conflict of interest related to this study.