Inflammatory response to Trichomonas vaginalis in the pathogenesis of prostatitis and benign prostatic hyperplasia
Article information
Abstract
Trichomonas vaginalis is a flagellated protozoan that causes trichomoniasis, a common nonviral sexually transmitted infection. T. vaginalis infection is asymptomatic in most infected men but can lead to chronic infection. The inflammatory response to chronic T. vaginalis infection may contribute to prostatic diseases, such as prostatitis and benign prostatic hyperplasia (BPH); however, studies on the relationship between T. vaginalis infection and prostate diseases are scarce. In this review, we discuss evidence from our studies on the involvement of T. vaginalis in the pathogenesis of prostate diseases, such as prostatitis and BPH. Studies of prostatitis have demonstrated that the attachment of T. vaginalis trophozoite to prostate epithelial cells (PECs) induces inflammatory cytokine production and inflammatory cell migration, leading to prostatitis. T. vaginalis also causes pathological changes, such as inflammatory cell infiltration, acinar changes, interstitial fibrosis, and mast cell infiltration, in prostate tissues of infected rats. Thus, T. vaginalis is considered an infectious agent that triggers prostatitis. Meanwhile, studies of prostatic hyperplasia revealed that mast cells activated by T. vaginalis-infected prostate cells secreted inflammatory mediators, such as β-hexosaminidase and tryptase, which promoted proliferation of prostate stromal cell (PSC). Moreover, interleukin-6 produced by proliferating PSCs induced the multiplication of BPH-1 epithelial cells as a result of stromal–epithelial interaction, suggesting that the proliferation of T. vaginalis-infected prostate cells can be induced through crosstalk with mast cells. These collective findings suggest that T. vaginalis contributes to the progression of prostatitis and prostatic hyperplasia by creating an inflammatory microenvironment involving PECs and PSCs.
Introduction
Trichomonas vaginalis is a flagellated protozoan belonging to the subphylum Mastigophora. Its total length, including the oval body, flagella, and axostyle, averages approximately 26 μm (21–32 μm). The body of T. vaginalis measures 9.5×6.8 μm, and the undulating membrane extends 3/4th of its body length [1]. T. vaginalis infects the urogenital system, causing trichomoniasis, and is the most common sexually transmitted protozoan. According to the World Health Organization estimates, the incidence of T. vaginalis infection is over 142 million cases [2]. In men, trichomoniasis causes pain during urination and ejaculation. Moreover, it increases the frequency of urination and results in a thin, white discharge from the penis as well as soreness, swelling, and redness around the head of the penis or foreskin. However, most T. vaginalis infections are asymptomatic, remain undiagnosed and untreated, and are thought to result in chronic persistent prostatic infection. In addition, trichomoniasis is a risk factor for human immunodeficiency virus transmission [3].
The prostate is a male urogenital organ located at the base of the bladder surrounding the urethra. It consists of 3 glandular zones, including the central, peripheral, and transition zones, along with a nonglandular region of the anterior prostate composed of fibromuscular stroma, which provides structural support [4]. The glandular tissue contributes to reproductive function by secreting zinc, citrate, and proteins, such as prostatic acid phosphatase, β-microsemiprotein, and prostatic-specific antigen, into the seminal fluid [5,6], whereas the fibromuscular tissue aids in the compartmentalization of fluids during urination and ejaculation.
Sfanos et al. [7] have suggested the existence of barrier functions and antimicrobial defenses in the prostate; epithelial cells in the prostate express pathogen pattern recognition receptors and Toll-like receptor 4 (TLR4), which may serve as defenses against prostate infections [8,9]. In addition, the prostatic fluid contains several antimicrobial proteins, such as lactoferrin, defensins, immunoglobulins, and complement proteins, as well as high levels of free zinc and citrate [10–12]. The strong antimicrobial activities of prostatic secretions may be important in the protection of the male genitourinary tract from ascending infections and sperm transiting into the female genital tract.
While microaerophilic T. vaginalis survive well in the vagina, which is essentially a hollow cavity, their survival becomes increasingly difficult once they reach the prostate, which contains antimicrobial proteins as well as high concentrations of zinc and citrate. Nevertheless, Gardner et al. [13] identified Tvs in the prostatic urethra, glandular lumina, submucosa, and stroma of 5 prostate glands obtained at autopsy via immunoperoxidase staining. The foci of nonspecific acute and chronic inflammation as well as intraepithelial vacuolization have also been reported to be associated with Tv infection. Moreover, Mitteregger detected Tv DNA in 34% of the prostate tissues obtained from 86 patients with benign prostatic hyperplasia (BPH), indicating that Tv is chronically present in BPH tissue and may influence prostate growth [14]. In addition, Iqbal et al. [15] detected T. vaginalis DNA and antigen in the prostate tissues of 42 (25%) and 37 (22%) patients with BPH, respectively.
Chronic infection with T. vaginalis may affect the development of prostatic hyperplasia as well as prostatitis. In this review, we discuss the hypothesis that T. vaginalis is involved in the initiation and/or progression of prostate diseases based on the results of both in vitro and in vivo experiments.
Prostatitis
Prostatitis is the most common urological diagnosis in men below 50 years of age, accounting for 8% of all office visits to urologists [16]. The most frequently diagnosed form of prostatitis is chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS). However, despite its high prevalence, prostatitis remains poorly understood, and the majority of diagnosed cases are of unknown etiology. Predisposing factors for CP/CPPS include heredity, infection, voiding abnormalities, hormone imbalance, intraprostatic reflux, immunological/allergic triggers, and psychological factors. Infection is responsible for 71–74% of chronic prostatitis cases, with 11–19% cases resulting from trichomoniasis infection [17,18]. In Korea, the prevalence of T. vaginalis infection, as determined via polymerase chain reaction, is 4 and 21% in patients with chronic prostatitis/urethritis and prostatic hyperplasia, respectively [19,20]. Despite these high prevalence rates, experimental induction of prostatic infection by T. vaginalis has not been attempted; however, it was determined whether exposure to T. vaginalis could induce an inflammatory response in normal prostate epithelial cells (PECs).
Adhesion of T. vaginalis to PECs and cytotoxic consequences
Alderete et al. [21,22] reported that the adhesion of T. vaginalis to mucosal cells is the first and essential step toward infection. Electron microscopy studies revealed that in vitro-grown T. vaginalis cells with a typical globular shape rapidly transform into flat and amoeboid cells upon contact with vaginal epithelial cells (VECs), maximizing the area of adhesion [23,24]. In contrast, little is known about the adherence of T. vaginalis to PECs. T. vaginalis stained with a CellTrackerOrange fluorescent probe was incubated with PECs (cell line RWPE-1) and observed under a fluorescence microscope. When PECs were incubated with T. vaginalis at ratios of 1:0.4 to 1:4, the adhesion of T. vaginalis began after 30 min and increased continuously for 24 h. When the number of trichomonads increased, the rate of adhesion to PECs also concomitantly increased [25]. At the RWPE-1: T. vaginalis ratio of 1:0.4, cytotoxic damage to PECs was evident at 9 h, whereas at the ratio of 1:4, it was observed at 3 h. Epithelial–mesenchymal transition (EMT) was confirmed by decreased E-cadherin expression and increased vimentin expression at 24 h [25,26], which was similar to that observed in type 2 EMT associated with wound healing and tissue regeneration.
When T. vaginalis attaches to PECs, it stimulates the production of IL-1β, IL-6, CCL2, and CXCL8 by activating ROS, ERK, and NF-κB signaling. Also, it has been reported that a PEC-conditioned medium containing these cytokines stimulates the migration of human neutrophils and monocytes. These data suggest that T. vaginalis induces an inflammatory response in PECs [27]. Because IL-1β and IL-6 play important roles in the progression of prostatic diseases, the signaling pathways involved in the production of these cytokines during T. vaginalis infection of PECs were examined.
Involvement of the NLRP3 inflammasome in IL-1β production by T. vaginalis-infected PECs
The NLRP3 inflammasome is involved in the production of IL-1β, a mediator of prostate inflammation associated with BPH, chronic prostatitis, and chronic pelvic pain syndrome [28,29]. The secretion of IL-1β is tightly regulated [30]. First, pro-IL-1β is produced following the activation of pattern recognition receptors. This precursor is subsequently cleaved into a mature form by the proinflammatory cysteine protease caspase-1. Caspase-1 is activated in response to infection or tissue damage and is modulated by a macromolecular protein complex known as the inflammasome, which consists of a NOD-like receptor (NLR) family member, an adaptor protein, and an inactive caspase-1 precursor [31]. The NLRP3 inflammasome has been implicated in antibacterial, viral, fungal, and parasitic immune responses [32]. When PECs are infected with live T. vaginalis, the expression of ASC, NLRP3, and caspase-1, all of which are components of the NLRP3 inflammasome, increases. Conversely, IL-1β production is decreased through siRNA-targeting of NLRP3 and caspase-1, confirming the involvement of the NLRP3 inflammasome in IL-1β production [33].
Signaling pathway involved in IL-6 production by T. vaginalis-infected PECs
IL-6 is a well-known biomarker of chronic prostatic inflammation and a mediator of chronic inflammation in prostate cancer [34,35]. Sutcliffe et al. [36] suggested that IL-6 production in T. vaginalis-exposed PECs promotes prostate carcinogenesis through the expression of proto-oncogenes, such as PIM1, c-MYC, and HMGA1; however, nothing is known regarding the signal pathways involved in IL-6 production in response to T. vaginalis. Stimulation of PECs by T. vaginalis resulted in the increased production of IL-6; increased expression of TLR2, TLR4, MAPKs, NF-κB, and JAK2/STAT3; and elevated levels of ROS. The inhibition of TLR2 or TLR4 reduced the production of IL-6 as well as the expression of other factors, and inhibitors of ROS, MAPKs, NF-κB, and JAK reduced IL-6 production. PECs stimulated with T. vaginalis underwent EMT, which was suppressed by the inhibitors of JAK or NF-κB. That is, T. vaginalis stimulates PECs to induce IL-6 production by activating TLR2/4, ROS, MAPKs, NF-κB, AP-1, JAK2/STAT3, and EMT via the NF-κB and JAK/STAT3 signaling pathways. Therefore, T. vaginalis may alter the prostate tumor microenvironment by inducing IL-6 production and EMT [26].
Prostate stromal inflammation in response to T. vaginalis
Prostatitis is caused by inflammatory responses that occurs not only in PECs but also in stromal cells. T. vaginalis has been detected in the submucosal layer and stroma of the prostate through immunoperoxidase staining [13]. The stromal compartment of the prostate primarily consists of smooth muscle cells, fibroblasts, macrophages, endothelial cells, and other immune cells [37]. The WPMY-1 cell line, isolated from normal human prostate stroma, is a myofibroblast line immortalized with SV40 large T antigen. Myofibroblasts play an important role in wound contraction in fibrotic diseases as an intermediate cell type between fibroblast and smooth muscle cell. Moreover, myofibroblasts are the dominant cell type associated with prostatic hypertrophy and contribute to prostate cancer progression and metastasis [38].
Previous studies showed that when RWPE-1 cells were incubated with T. vaginalis, cell viability at 24 h decreased concomitantly with an increase in the number of trichomonads, confirming that T. vaginalis is cytotoxic to RWPE-1 [25,26]. Sfanos et al. [7] suggested that epithelial cell death and consequent barrier function disruption lead to infiltration of stromal compartments by commensal or pathogenic organisms. It has been hypothesized that T. vaginalis penetrates the epithelium, enters the stroma, and induces an inflammatory response. Therefore, it was determined whether T. vaginalis induces an inflammatory response in stromal cells. Incubation of WPMY-1 with live T. vaginalis increased the expression of the inflammatory chemokines CXCL8 and CCL2. In addition, TLR4, ROS, MAPK, and NF-κB expression increased, whereas the inhibitors of TLR4, ROS, MAPKs, and NF-κB decreased CXCL8 and CCL2 production. Conditioned medium derived from WPMY-1 cells incubated with T. vaginalis stimulated the migration of human neutrophils and monocytes, suggesting that T. vaginalis increases CXCL8 and CCL2 production by human prostate stromal cells by activating TLR4, ROS, MAPKs and NF-κB. This, in turn, attracts neutrophils and monocytes and results in an inflammatory response [39].
T. vaginalis infection causes rat prostatitis
The in vitro inflammatory responses induced by T. vaginalis in PECs and stromal cells prompted us to determine whether T. vaginalis causes prostatitis in an animal model. To mimic the route of T. vaginalis infection in humans, T. vaginalis was injected through the urethras of Wistar rats. T. vaginalis trophozoites were observed in the prostate acini of the injected rats via immunohistochemistry. The prostate tissues exhibited elevated pathological scores, and 83% (5/6 samples) and 100% (6/6 samples) of the ventral and dorsolateral lobes (n=6), respectively, were inflamed. Pathological changes, such as inflammatory cell infiltration, acinar changes, and interstitial fibrosis, were observed. The inflammation was more severe in the dorsolateral lobes than in the ventral lobes. Moreover, infiltration and degranulation of mast cells were observed at higher rates in the prostate sections of the T. vaginalis-infected rats, and the prostate tissues of the injected rats had increased CCL2 levels. These findings provided the first evidence that T. vaginalis infection in rats causes prostatitis [40].
Summary of prostatitis experiments
Taken together, the above results indicate that the mechanism by which T. vaginalis causes prostatitis may occur as follows: T. vaginalis causes prostatitis by attaching to prostate cells and inducing proinflammatory cytokine production, inflammatory cell migration, and EMT. In animal experiments, injection of T. vaginalis into rats via the urethra caused prostatitis as evidenced by various pathologic changes, including inflammatory cell infiltration, acinar changes, interstitial fibrosis, mast cell infiltration, and increased CCL2 production. These in vitro and in vivo experimental evidence demonstrate that T. vaginalis is an infectious agent causing prostatitis (Fig. 1).
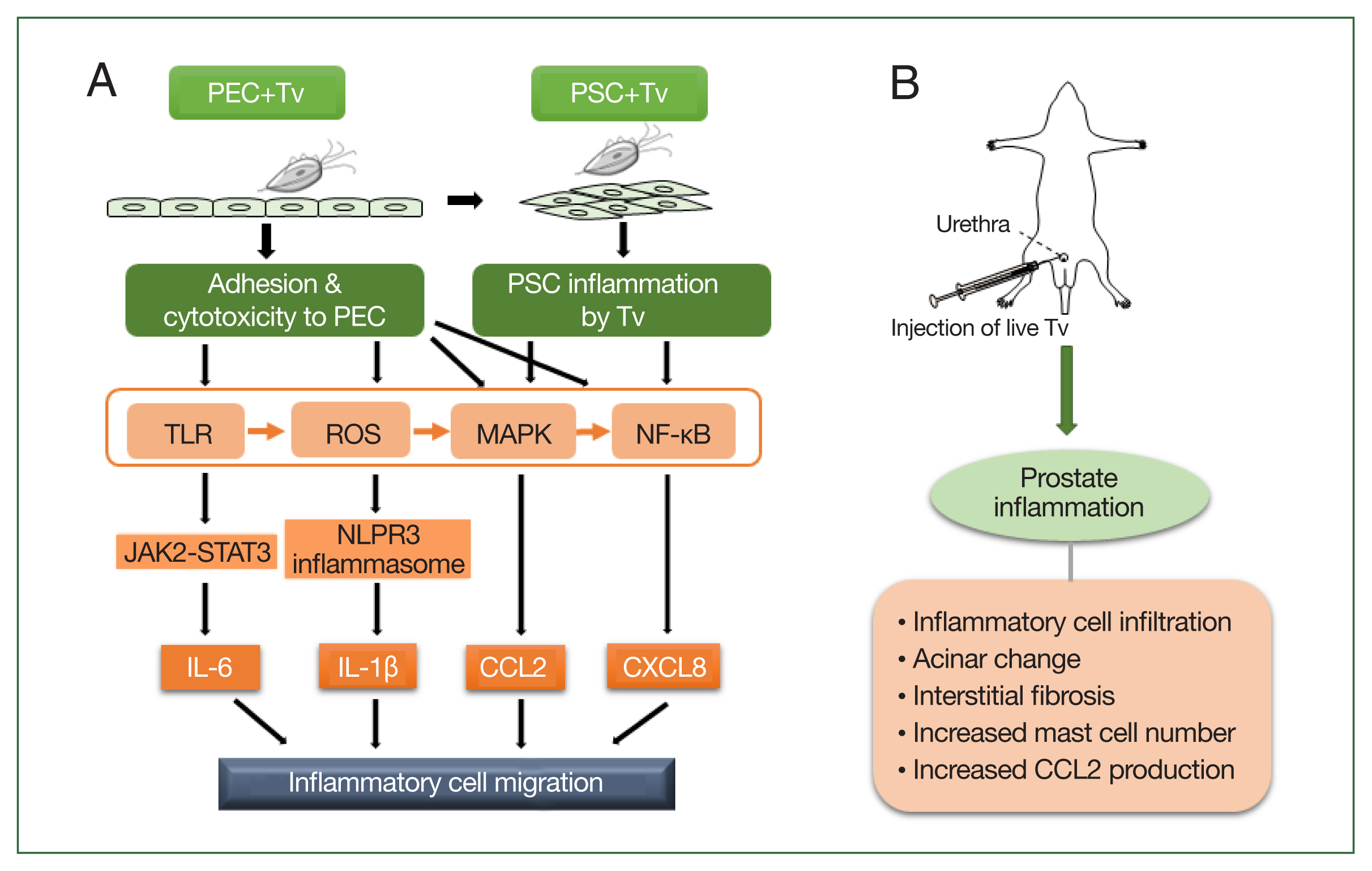
Schematic diagram of the induction of prostatitis by Trichomonas vaginalis (Tv). (A) Tv attaches to prostate epithelial cells (PECs) and causes an inflammatory response, resulting in the increased production of cytokines, such as IL-1β, IL-6, CXCL8, and CCL2. In addition, Tv causes cytotoxic damage to epithelial cells, thereby infecting prostate stromal cells (PSCs) and triggering an inflammatory response that induces the migration of inflammatory cells. TLR, ROS, MAPK, and NF-kB are generally involved in cytokine production, and in particular, the NLRP3 inflammasome and JAK2-STAT3 are implicated in IL-1β and IL-6 production, respectively. (B) Injection of live Tv through the rat urethra induces prostatitis.
Prostate Hyperplasia
Benign prostate hyperplasia (BPH)
BPH results in the benign enlargement of the prostate gland due to the unregulated hyperplastic growth of the epithelial and fibromuscular tissues of the transition zone and periurethral area [41]. BPH is an immune-mediated inflammatory disease, with persistent prostatic inflammation as a key factor in its development and progression [42,43]. An inflammatory reaction may be triggered by several factors, including bacterial infection (e.g., Escherichia coli), viruses (e.g., human papilloma virus, herpes simplex virus type 2, and cytomegalovirus), sexually transmitted organisms (e.g., gonorrhea and chlamydia), hormones, dietary factors, autoimmune response, and urinary reflux into the prostate collection ducts [42,44–46]. The inflammatory response induced by these factors may cause T-lymphocyte infiltration, cytokine production, growth factor expression, local hypoxia with angiogenesis, tissue damage with abnormal wound healing and stromal and epithelial cell proliferation, and eventually BPH [42,45].
Several sexually transmitted micro organisms, such as Neisseria gonorrhoeae, Chlamydia trachomatis, and T. vaginalis, are known to cause chronic inflammation of the prostate tissue [47]. However, in a study investigating the association between BPH (or lower urinary tract symptom-related outcomes) and antibodies against various sexually transmitted infections (e.g., C. trachomatis, T. vaginalis, human papilloma virus, herpes simplex virus, cytomegalovirus infection, gonorrhea, and syphilis), only T. vaginalis infection was found to be linked to BPH [48]. Moreover, T. vaginalis DNA was detected in the prostate tissue and urine of patients with BPH, suggesting that it acts as an initial stimulus for an inflammatory response [14,15,19]. In addition, Twu et al. [48] reported that T. vaginalis macrophage migration inhibitory factor induced an inflammatory response, prostate cell growth, and invasiveness in BPH-1 and prostate cancer cell lines.
To date, there have been few studies regarding prostate growth induced by the inflammatory response of prostate cells infected with live T. vaginalis. Claus et al. [50] reported that the proliferation rates of prostate epithelium and stroma increased by approximately 9-fold and 37-fold, respectively, in BPH compared with the normal prostate and that prostatic stromal hyperproliferation was an important feature of BPH pathogenesis. We focused on stromal hyperplasia in the experimental studies of prostatic hyperplasia.
We hypothesized that the crosstalk between prostatic cells inflamed by T. vaginalis infection and migrating inflammatory cells was involved in the proliferation of prostatic cells. During inflammatory reaction in prostate tissue, T lymphocytes are the most common infiltrating cells. B lymphocytes and macrophages are also known to infiltrate [42,44]. However, clinical studies have suggested that mast cells play an important role in the development and persistence of inflammation in BPH associated with the lower urinary tract [51–53]. In our prostate proliferation studies, we focused on mast cells based on reports that they are involved in the inflammatory response to T. vaginalis infection. Mast cells were preferentially detected in cervical smears from women with trichomoniasis compared with smears from patients with vaginitis caused by other pathogens [54]. T. vaginalis (live trophozoites, excretory-secretory products [ESP]) was confirmed to induce an inflammatory response by migrating and activating mast cells, which resulted in the production of histamine and TNF-α [55]. When VECs were cocultured with T. vaginalis, VEC-produced inflammatory mediators activated and attracted mast cells and stimulated them to induce neutrophil migration [56]. In addition, the number of mast cells and activated degranulated mast cells increased during prostatitis in T. vaginalis-infected rats [40].
Mast cells are important cellular sensors and regulators of inflammation, fibrosis, and smooth muscle cell contraction [53,57–59]. Activated mast cells release various factors, such as chymase, tryptases, and proteases, which can interact with the local tissue environment and lead to tissue fibrosis, repair, and remodeling [53]. Conversely, little is known regarding the role of mast cells in prostate proliferation resulting from T. vaginalis infection.
Proliferation of stromal cells is induced by crosstalk between T. vaginalis-infected BPH epithelial cells and mast cells
T. vaginalis-infected BPH epithelial cells have been reported to produce cytokines, such as CXCL8, CCL2, IL-1β, and IL-6, through the ROS, MAPK, and NF-κB signaling pathways. Inflammatory mediators, including these cytokines, stimulate the migration of monocytes and mast cells. When mast cells are incubated with trichomonad-conditioned medium, the activated mast cells produce β-hexosaminidase and CXCL8 and induce PSC proliferation. Proliferation was decreased by inhibiting CXCR1 (receptor for CXCL8) and CCR2 (receptor for CCL2), indicating that CXCL8 and CCL2 stimulate PSC proliferation by binding to CXCR1 and CCR2, respectively. This study indicated that T. vaginalis-infected BPH-1 cells activate mast cells, which release inflammatory mediators that induce PSC proliferation [60,61].
Induction of PSC proliferation by tryptase derived from activated mast cells
Tryptase is the most abundant serine proteinase found in the secretory granules of mast cells and has been reported to induce human lung fibroblast migration, myoblast proliferation, and cardiac fibroblast activation via protease-activated receptor 2 (PAR2) [62–64]. PAR2 is a G-protein-coupled receptor activated through proteolytic cleavage by serine proteases, including tryptase. PAR2 may be involved in tissue remodeling by inducing fibroblast migration, differentiation, and extracellular matrix production [65]. Roman et al. [66] suggested that PAR2 activation in the prostate contributes to the development of lower urinary tract dysfunction through proinflammatory as well as profibrotic pathways. Furthermore, activation of the tryptase–PAR2 axis is important for the onset of fibrosis.
Studies have examined whether tryptase released from mast cells activated by T. vaginalis-infected prostate stromal cells (PSC) promotes the proliferation of PSCs via protease-activated receptor 2 (PAR2). Inflammatory mediators derived from T. vaginalis-infected PSCs induced mast cell migration, tryptase production, and PSC proliferation. Signaling molecules involved in the tryptase–PAR2 pathway (e.g., PAR2, p-ERK, COX-2, 15d-PGJ2, and PPARγ) were increased, and the inhibition of tryptase and signaling molecules inhibited proliferation. These results indicate that the interaction between T. vaginalis-infected PSCs and mast cells induces the proliferation of PSCs via the tryptase–PAR2 pathway [67].
Induction of PEC proliferation through stromal–epithelial cell interaction
According to the American Urological Association, BPH is defined as a histologic diagnosis and refers to the proliferation of smooth muscle and epithelial cells within the prostatic transition zone [68]. Although there are reports regarding the proliferation of prostate stromal cells, only limited information is available on the proliferation of PECs.
Stromal–epithelial interactions play an important regulatory role in the development of the prostate and in the maintenance of the adult prostate in health and disease [69]. Siejka et al. [70] reported that bidirectional stromal–epithelial interactions occur in the prostate gland. When the supernatants of prostate stromal cells (or BPH epithelial cells) were added to BPH epithelial cells (or prostate stromal cells), proliferation was increased. In BPH tissues, the ratio of stromal to epithelial cells reached a value of 5.
As discussed in Proliferation of stromal cells is induced by crosstalk between T. vaginalis-infected BPH epithelial cells and mast cells, the proliferation of prostate stromal cells is induced by BPH epithelial cells in response to T. vaginalis infection through crosstalk with mast cells. Subsequently, the effect of IL-6 released by proliferating stromal cells on the induction of BPH epithelial cells was determined. When culture supernatants of proliferating prostate stromal cells were added to BPH epithelial cells, the latter multiplied and cyclin D1, FGF2, and Bcl-2 expression increased. Blocking the IL-6 signaling pathway with anti-IL-6R antibody or a JAK1/2 inhibitor inhibited the proliferation of BPH epithelial cells and reduced the expression of IL-6, IL-6R, and STAT3. EMT was also detected in proliferating BPH epithelial cells, indicating that IL-6 released from proliferating prostate stromal cells induced by BPH epithelial cells infected with T. vaginalis promoted BPH epithelial cell multiplication [71]. This indicates that the inflammatory microenvironment of prostate stromal cells resulting from T. vaginalis infection promotes the proliferation of PECs through stromal–epithelial interaction.
EMT is a complex process that involves the transformation of epithelial cells into stromal cells. It is integral to development, wound healing, and stem cell behavior, and it contributes to fibrosis and cancer progression [72,73]. Alonso-Magdalena et al. [74] analyzed prostate samples from 16 patients with BPH. BPH is not a disease of the prostate stroma but is the accumulation of mesenchymal-like cells derived from the prostate epithelium, suggesting that EMT plays an important role in the development of BPH. Shi et al. [75] and Hu et al. [76] reported that estradiol and TGF-B1/Smad induce EMT in BPH epithelial cells. Thus, EMT in BPH epithelial cells proliferating in response to IL-6 produced by T. vaginalis-infected stromal cells is involved in the pathogenesis of BPH.
Involvement of adipocyte leptin in T. vaginalis-induced proliferation of prostate cells
BPH may be linked to obesity as abdominal obesity and serum leptin levels were found to be associated with prostate growth in a retrospective cohort study in Korea [77]. Male obesity has also been linked to increased severity of lower urinary tract symptoms (LUTS) in men affected by BPH [78]. Adipocytes, also known as lipocytes or fat cells, are the main components of adipose tissue that specialize in storing energy as fat. Adipokines produced by adipose tissue affect metabolic processes and mediate inflammation, cell proliferation, and angiogenesis [79]. Leptin is an adipokine originally identified as a key molecule regulating food intake and body weight. It interacts with other hormones and energy regulators to mediate the effects of insulin, glucagon, insulin-like growth factor, growth hormone, glucocorticoids, cytokines, and metabolites [80]. Leptin also stimulates the growth of prostate, breast, lung, ovarian, and pancreatic cancer cells [81]. Prostate cells proliferate when leptin is administered together with estrogen; however, reports of prostate cell proliferation in response to leptin alone are rare [82].
A study was performed to determine whether T. vaginalis-infected BPH epithelial cells (BPH-1 cells) induced the proliferation of prostate cells through a leptin signaling pathway. BPH-1 cells incubated with live T. vaginalis released proinflammatory cytokines, and conditioned medium from these cells stimulated adipocyte migration. When PSCs and BPH-1 cells were incubated with adipocyte-conditioned medium containing leptin (ATCM), their growth rates increased. In addition, the expression of the leptin receptor (known as OBR) and downstream signaling molecules, such as JAK2/STAT3, Notch1/Jagged1, and survivin, increased. Moreover, OBR blocking reduced proliferation and the expression of leptin signaling molecules in response to ATCM. These findings indicate that inflamed BPH-1 cells infected with T. vaginalis promote prostate cell proliferation through leptin–OBR signaling. Therefore, it is likely that T. vaginalis contributes to prostate enlargement in BPH via adipocyte leptin release as a result of prostate inflammation [83].
Summary of prostate cell proliferation experiments
Based on the above results, the mechanism by which T. vaginalis induces prostate cell proliferation may be as follows: when T. vaginalis interacts with prostate cells, it triggers an inflammatory response, and migratory mast cells induce PSC proliferation by secreting inflammatory mediators or producing tryptase. Subsequently, the proliferating PSCs produce IL-6, which stimulates BPH-1 epithelial cell proliferation. These findings indicate that bidirectional stromal–epithelial interactions are responsible for PEC proliferation. In addition, when BPH-1 cells are infected with T. vaginalis, adipocytes migrate and produce leptin, which results in the proliferation of prostate cells through the leptin–OBR signaling pathway (Fig. 2). Taken together, it appears that T. vaginalis causes proliferation of the prostate via the infiltration of inflammatory cells, including mast cells and adipocytes.
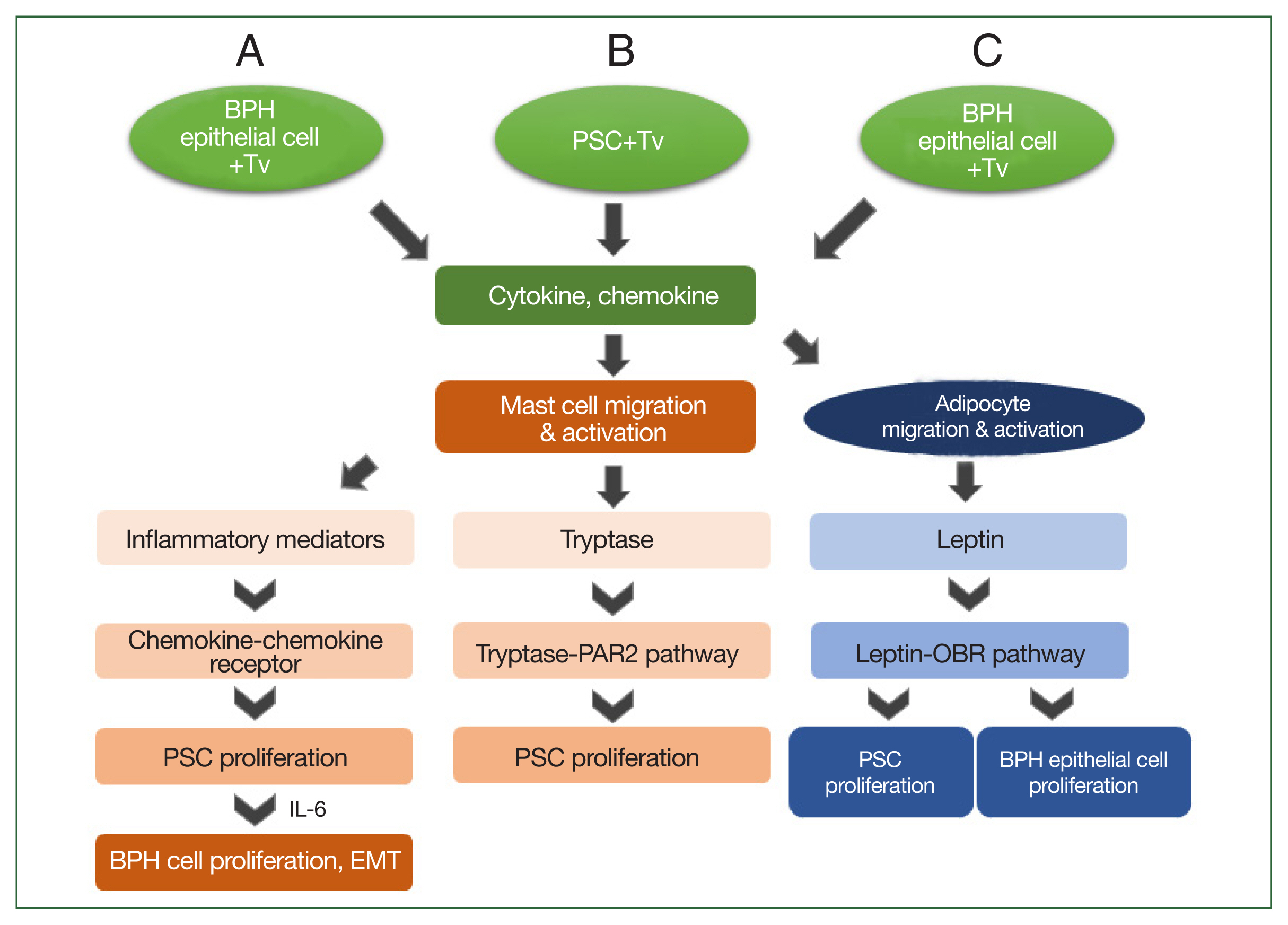
Schematic diagram of the induction of benign prostatic hyperplasia (BPH) by Trichomonas vaginalis (Tv). When BPH-1 epithelial cells and prostate stromal cells (PSCs) interact with Tv, they produce cytokines and chemokines that activate mast cells or adipocytes and stimulate their migration. (A) For BPH-1 cells, chemokines from activated mast cells bind to a chemokine receptor on PSCs to induce their proliferation. Subsequently, the proliferating PSCs produce IL-6, which results in BPH-1 epithelial cell proliferation. (B) Tryptase released from mast cells activated by Tv-infected PSCs promotes the proliferation of PSCs through the tryptase–protease-activated receptor 2 (PAR2) signaling pathway. (C) Cytokines and chemokines released by Tv-infected BPH-1 cells trigger adipocyte migration and activation. Leptin produced by activated adipocytes induces prostate cell proliferation through the leptin–OBR signaling pathway.
Conclusion
When T. vaginalis interacts with prostate cells, such as PECs, prostate stromal cells, or BPH epithelial cells, inflammatory mediators such as cytokines are produced to create an inflammatory microenvironment. In addition, the inflammatory mediators released from activated inflammatory cells, including migratory mast cells and adipocytes, amplify the inflammatory response via cell–cell interactions, leading to prostatitis or BPH. Although this review highlights some pathogen-induced prostate inflammatory responses, more studies in this field are needed.
Acknowledgments
We thank Min-Young Seo, Su-Jeong Lim, Na-Young Gu, Sang-Su Kim, Hyo-Yeoung Chung, Dr. Kyu-Shik Kim, Dr. Ik-Hwan Han, Dr. Jung-Hyun Kim, and the late Dr. Ki-Seok Jang for participating in the experiments. Also we would like to thank Professor Soon-Jung Park for revising this review.
Notes
Author contributions
Conceptualization: Ryu JS
Data curation: Kim JH
Investigation: Han IH, Kim JH, Ryu JS
Methodology: Han IH, Kim JH
Supervision: Ryu JS
Writing – original draft: Han IH, Ryu JS
The authors declare no conflict of interest related to this study.
