Cited By
Citations to this article as recorded by

Toxoplasma gondii infection and the risk of adult glioma in two prospective studies
James M. Hodge, Anna E. Coghill, Youngchul Kim, Noemi Bender, Stephanie A. Smith‐Warner, Susan Gapstur, Lauren R. Teras, Tom K. Grimsrud, Tim Waterboer, Kathleen M. Egan
International Journal of Cancer.2021; 148(10): 2449.
CrossRef Toxoplasma gondii infection in patients with brain tumors in Southern Iran: a case-control study
Qasem Asgari, Farshad Rajabi, Fataneh Sajadian, Mohammad Saleh Bahreini, Nasir Arefkhah
Journal of Parasitic Diseases.2023; 47(2): 291.
CrossRef Increased risk of Toxoplasma gondii infection in cancer patients: A meta-analysis of current evidence based on case-control study
Lei Liu, Peng Wang, Jing Xu, Chao-Ming Xia
Toxoplasma gondii: Seroprevalence and association with childhood brain tumors in Egypt
Marwa M. Hamouda, Amany S. El-Saied, Ahmed Zaher, Amr Farid Khalil, Ayat A. ElBlihy, Nairmen Nabih, Samar N. El-Beshbishi
Acta Tropica.2024; 251: 107123.
CrossRef Toxoplasma gondii infection/exposure and the risk of brain tumors: A systematic review and meta-analysis
Ali Abdollahi, Iman Razavian, Elnaz Razavian, Sahar Ghodsian, Mustafa Almukhtar, Elika Marhoommirzabak, Behnam Sartip, Hamid Parsa, Ali Rostami
Cancer Epidemiology.2022; 77: 102119.
CrossRef Seroprevalence of Toxoplasma gondii infection in cancer patients: A systematic review and meta-analysis
Davood Anvari, Mehdi Sharif, Shahabeddin Sarvi, Sargis A. Aghayan, Shirzad Gholami, Abdol Sattar Pagheh, Seyed Abdollah Hosseini, Reza Saberi, Tooran Nayeri Chegeni, Zahra Hosseininejad, Ahmad Daryani
Microbial Pathogenesis.2019; 129: 30.
CrossRef Serology for Toxoplasma in Immunocompromised Patients: Still Useful?
Damien Dupont, Hélène Fricker-Hidalgo, Marie-Pierre Brenier-Pinchart, Cécile Garnaud, Martine Wallon, Hervé Pelloux
Trends in Parasitology.2021; 37(3): 205.
CrossRef Puzzling and ambivalent roles of malarial infections in cancer development and progression
ERIC FAURE
Parasitology.2016; 143(14): 1811.
CrossRef Seroprevalence and risk factors of Toxoplasma gondii infection in oral cancer patients in China: a case–control prospective study
N. Zhou, X. Y. Zhang, Y. X. Li, L. Wang, L. L. Wang, W. Cong
Epidemiology and Infection.2018; 146(15): 1891.
CrossRef Toxoplasma gondii and Rickettsia spp. in ticks collected from migratory birds in the Republic of Korea
A.-Tai Truong, Mi-Sun Yoo, Subin Min, Ji-Yeon Lim, Hyun-Ji Seo, Heung-Chul Kim, Sung-Tae Chong, Terry A. Klein, Chang-uk Park, Sook-Young Cho, Chang-Yong Choi, Young-Soo Kwon, Miran Kim, Soon-Seek Yoon, Yun Sang Cho
Whorling‐sclerosing meningioma invading skull bone and subcutaneous tissue with an incidental toxoplasmosis: A case report
Zeina Alabbas, Yamani Tamer, Mhammad Jneidi, Rana Issa
Neuropathology.2023; 43(3): 262.
CrossRef Relationship between parasites and lung cancer: Unveiling the link
Mariam T. El Khadrawe, Nahla El Skhawy, Maha M. Eissa
Tropical Medicine & International Health.2025;[Epub]
CrossRef Differentiating Cerebral Toxoplasmosis and Tumor Recurrence by Thallium-201 Single-Photon Emission Computerized Tomography in a 28-Year-Old Female with Astrocytoma
Mia Patrice Dela Vega, Paul Vincent Opinaldo, Julette Marie Batara
Case Reports in Oncology.2023; : 372.
CrossRef Molecular Detection of Toxoplasma Gondii in Haemaphysalis Ticks in Korea
Ju Yeong Kim, You Shine Kwak, In-Yong Lee, Tai-Soon Yong
The Korean Journal of Parasitology.2020; 58(3): 327.
CrossRef Imiquimod Targets Toxoplasmosis Through Modulating Host Toll-Like Receptor-MyD88 Signaling
Maguy Hamie, Rania Najm, Carine Deleuze-Masquefa, Pierre Antoine Bonnet, Jean-François Dubremetz, Marwan El Sabban, Hiba El Hajj
Frontiers in Immunology.2021;[Epub]
CrossRef Infratentorial Stereotactic Biopsy of Brainstem and Cerebellar Lesions
Jacek Furtak, Paulina Śledzińska, Marek G. Bebyn, Tadeusz Szylberg, Stanisław Krajewski, Marcin Birski, Marek Harat
Brain Sciences.2021; 11(11): 1432.
CrossRef Comprehensive Overview of Toxoplasma gondii-Induced and Associated Diseases
Darine Daher, Ahmad Shaghlil, Eyad Sobh, Maguy Hamie, Malika Elhage Hassan, Mohamad Bahij Moumneh, Shaymaa Itani, Rana El Hajj, Lina Tawk, Marwan El Sabban, Hiba El Hajj
Abstract
Toxoplasma gondii is an intracellular protozoan that can modulate the environment of the infected host. An unfavorable environment modulated by T. gondii in the brain includes tumor microenvironment. Literature has suggested that T. gondii infection is associated with development of brain tumors. However, in Korea, epidemiological data regarding this correlation have been scarce. In this study, in order to investigate the relationship between T. gondii infection and brain tumor development, we investigated the seroprevalence of T. gondii among 93 confirmed brain tumor patients (various histological types, including meningioma and astrocytoma) in Korea using ELISA. The results revealed that T. gondii seropositivity among brain tumor patients (18.3%) was significantly (P<0.05) higher compared with that of healthy controls (8.6%). The seropositivity of brain tumor patients showed a significant age-tendency, i.e., higher in younger age group, compared with age-matched healthy controls (P<0.05). In conclusion, this study supports the close relationship between T. gondii infection and incidence of brain tumors.
Key words: Toxoplasma gondii, seroprevalence, brain tumor, ELISA
More than 14.1 million common cancer cases are estimated around the world in 2012, and this number is anticipated to increase to 24.0 million by 2035 [
1]. About 256,000 central nervous system tumors (1.8% of all tumors) are diagnosed each year worldwide [
1]. Previous studies have found that risk factors for brain tumors include diverse chemical products, family history, and ionizing radiation from therapeutic and diagnostic devices in the head [
2]. However, the exact causes of these malignancies are yet unclear. Approximately 20% of various malignancies worldwide are due to infectious agents, including viruses, bacteria, and parasites [
3,
4]. Infectious agents can interfere with the host cell genetic machinery, such as DNA repair and cell cycle, and can lead to chronic inflammation and immune system impairments [
5].
The infectious agents associated with human cancers are most commonly viral pathogens, including human papilloma virus and hepatitis B and C viruses [
6]. However, there are only a few studies on the association between parasites and human cancers. It has been suggested that several parasite species, namely,
Paragonimus westermani,
Plasmodium sp.,
Opisthorchis viverrini,
Clonorchis sinensis,
Schistosoma haematobium, and
Hymenolepis nana are related to development of various types of human cancers [
7,
8]. Many other species of parasites may also have potential roles in development of human cancers.
Toxoplasma gondii is an intracellular protozoan that can modulate the microenvironment of the infected host [
9].
T. gondii can invade vital organs, including the central nervous system (CNS); however, its infection in humans is usually mild and asymptomatic in immunocompetent individuals [
9]. In immunocompromised patients,
T. gondii infection may cause severe diseases in the brain, including fatal meningitis and encephalitis [
9]. In addition, a possible correlation was suggested between
T. gondii infection and brain tumor development [
10]. This suggestion has been supported by other authors [
11-
18]. The mechanisms of the brain tumor induction by
T. gondii need to be further studied. In this study, in order to estimate the possible relationship between
T. gondii infection and brain tumor development, we investigated the seroprevalence of
T. gondii among brain tumor patients diagnosed in Korea.
We used sera of 93 patients (44 men and 49 women) diagnosed with various types of brain tumors supplied by the Biobank of Chonnam National University Hwasun Hospital (Hwasun, Jeollanam-do Province, Korea), a member of the Korea Biobank Network. As the control group, sera of 93 randomly selected healthy volunteers (45 men and 48 women) who visited the Korea Association of Health Promotion for health check-up were included for the assay. The ages of the brain tumor patients were 18-82 years (52.9±14.8 years) and those of the healthy controls were 10-86 years (51.9±12.2 years). The sera and blood were stored at -80˚C until analyzed. This study protocol was approved by the Institutional Review Board of Seoul National University Hospital, Seoul, Korea (IRB no. E-1507-065-687). The purpose and procedures of this study were explained to all participants, and a written informed consent was obtained from each of them.
The process of preparation for
Toxoplasma lysate antigens (TLA) [
19] and the procedure of ELISA [
14] followed previous studies with slight modifications. Briefly, 96-well microtiter plates (Costar, Cambridge, Massachusetts, USA) coated with TLA were incubated at 4˚C overnight. After washing, each well was reacted with the test serum samples (1:100) at 37˚C for 1 hr, and horseradish peroxidase-conjugated goat anti-human IgG (1:10,000; Bethyl Laboratories, Montgomery, Texas, USA) was applied at 37˚C for 1 hr. After several washes, freshly prepared o-phenylenediamine dihydrochloride (Sigma-Aldrich, St. Louis, Illinois, USA) was added, and the reaction was stopped by adding 8 N H
2SO
4. IgG antibody titers were determined at the optical density of 490 nm. To analyze the risk factors for toxoplasmosis, the chi-square test was applied.
P<0.05 was considered statistically significant. Pearson’s chi-square and Fisher’s exact tests were used to investigate associations among qualitative categorical variables using SPSS (SPSS Inc., Chicago, Illinois, USA). All tests were 2-sided, and the level of significant difference was defined as
P<0.05.
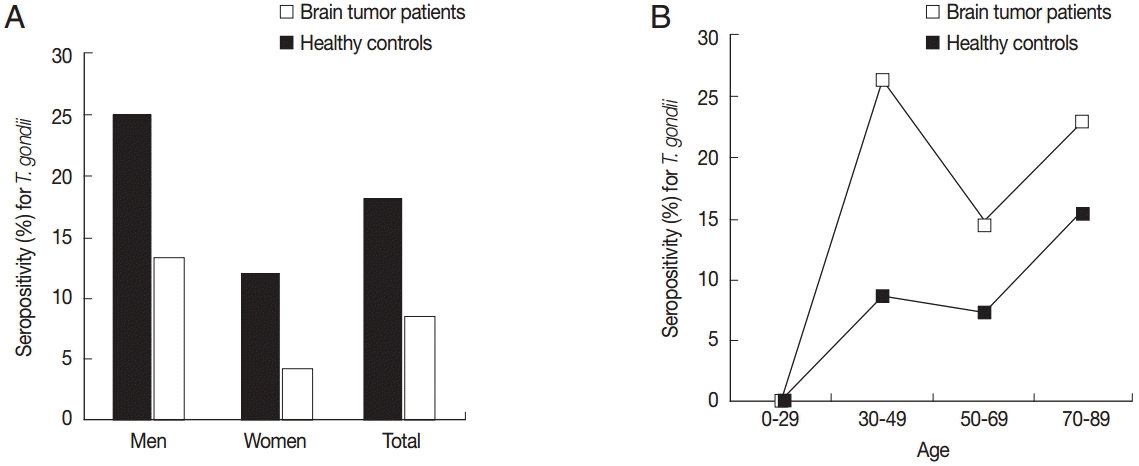
The IgG seropositive rate for
T. gondii, as analyzed by ELISA, was 18.3% (17/93) among patients with variable types of brain tumors (
Table 1). The seropositivity among brain tumor patients was significantly (
P<0.05) higher than the one among healthy persons, 8.6% (6/93 sera). In particular, patients with meningioma (41.7%;
P<0.05), metastatic carcinoma (30.0%;
P<0.05), astrocytoma (21.4%;
P>0.05), and glioblastoma (16.1%;
P>0.05) showed remarkably higher seropositivity for
T. gondii than healthy controls (
Table 1). The seropositivity was significantly (
P<0.05) higher in men than in women both in the brain tumor patients and healthy controls (
Fig. 1A). The age-seropositivity curve in healthy controls generally showed a steady increasing pattern according to increase of the age (
Fig. 1B), whereas in brain tumor patients the younger age group, for example, 30-49 years, revealed a significantly (
P<0.05) higher seropositivity compared with other age groups (
Fig. 1B).
Previous studies have shown that meningioma and astrocytoma were positively linked to high serum
T. gondii IgG antibody levels [
10,
13]. Similarly, our study showed higher prevalence of
Toxoplasma specific IgG in patients with meningioma (41.7%;
P<0.05) and astrocytoma (21.4%;
P>0.05) (
Table 1). Furthermore, in healthy controls, the seropositivity of
T. gondii was increased with age [
19,
20]. However, in our study, the seropositivity in brain tumor patients showed a different tendency; a significantly higher rate was observed in younger aged patients (30-49 years) with brain tumors, compared with age-matched healthy controls. These results support strongly the close relationship between
Toxoplasma infection and brain tumor incidence.
Since the 1960s, quite a number of studies [
10-
18] reported correlations between
T. gondii infection and brain tumor incidence (
Table 2). Various types of brain and multiple organ tumors, including glioma, acoustic neuroma, meningioma, Hodgkin’s lymphoma, multiple myeloma, and leukemia, have been reported to be positively associated with chronic
T. gondii infection (
Table 2). In this study, we also observed a high prevalence of
T. gondii infection in sera of various brain tumor patients in Korea. Especially, our study supports the previous finding that IgG seropositivity to
T. gondii is a risk factor for meningioma [
13]. These findings provide a strong need for a further study to establish the precise correlation between meningioma incidence and
T. gondii infection. However, results in this study have limitation to precisely explain the relationship between
T. gondii infection and brain tumor.
To evade host immune responses,
T. gondii parasites transform themselves into tissue cysts, and they are parasitic on various tissues, including the brain, heart, and skeletal muscle for lifetime of the host modulating the host immune responses [
21]. Furthermore, they can also modulate the cell cycle and apoptosis of the host cells for their proliferation [
17,
22]. Such behavioral pathogenesis of
T. gondii infection may be linked to the related hallmarks of tumor development. The unfavorable environment modulated by
T. gondii may be similar to appropriate precancerous conditions. Thus, we can presume that
T. gondii infection should help to develop brain tumors. Hosts with chronic
T. gondii infections are also vulnerable to the attack by other pathogens such as viral pathogens. If such viral pathogens are associated with brain tumor development, it can be another oncogenic effect of
T. gondii and may synergistically act as a carcinogen. In this respect, it is noteworthy that
Plasmodium falciparum and Epstein-Barr virus contributed synergistically to the formation of Burkitt’s lymphoma [
23].
Despite the studies on the association of
T. gondii and brain tumors, we cannot conclude whether chronic
T. gondii infection, with seropositivity, is involved in causing these brain tumors, or reversely brain tumor patients are at risk of recrudescence and dissemination of
T. gondii to undergo a chronic infection [
14]. Researches are also needed to determine whether
T. gondii itself acts as a carcinogen or takes the role of a creator of a precancerous environment to develop into brain tumors.