Abstract
In order to determine the status of Enterobius vermicularis infection among schoolchildren in suburban areas of Myanmar, 761 primary schoolchildren in 3 different townships around Yangon City were subjected to a survey using cello-tape anal swabs. The subjected schoolchildren were 383 boys and 378 girls who were 5-7 years of age. Only 1 anal swab was obtained from each child. The overall egg positive rate of E. vermicularis was 47.2% (359 positives), and sex difference was not remarkable (48.6% in boys and 45.8% in girls). However, the positive rate was the highest in South Dagon (54.6%) followed by Hlaing Thayar (43.8%) and North Dagon (34.8%). This difference was highly correlated with the living standards of the people in each township. Nucleotide sequence of the 5S rDNA from the eggs on the cello-tape (2 children) revealed 99.7% identity with that of E. vermicularis reported in GenBank. The results indicated that E. vermicularis infection is highly prevalent among primary schoolchildren around Yangon, Myanmar.
-
Key words: Enterobius vermicularis, enterobiasis, high prevalence, anal swab, schoolchildren, Myanmar
Intestinal helminthiases, in particular, soil-transmitted and contact-borne helminth infections are the most common parasitic infections in tropical and subtropical countries [
1]. No less than a billion people are infected with at least one species of intestinal helminth [
1]. The pinworm,
Enterobius vermicularis, is the most common helminth of humans and has a worldwide distribution including even developed countries such as Western Europe and United States. The estimated global population infected by the pinworm is about 4-28% [
1]. The common mode of pinworm transmission is ingestion of eggs either directly or indirectly through hands, bedding, clothing, and toilet seats [
2,
3]. Enterobiasis is commonly asymptomatic; however, children with high parasitic burdens have impairments in physical, intellectual, and cognitive development [
4].
Until present, there have been few documented reports on the prevalence of
E. vermicularis among people in Myanmar. A Thai research group reported that the egg positive rate of
E. vermicularis among 372 immigrant children from Myanmar who lived in Samut Sakhon Province, Thailand was 25.2% [
5]. No other reports are available. Thus, in the present study, we performed a small survey on
E. vermicularis infection targeting primary schoolchildren in 3 different townships located around Yangon City, Myanmar in 2014.
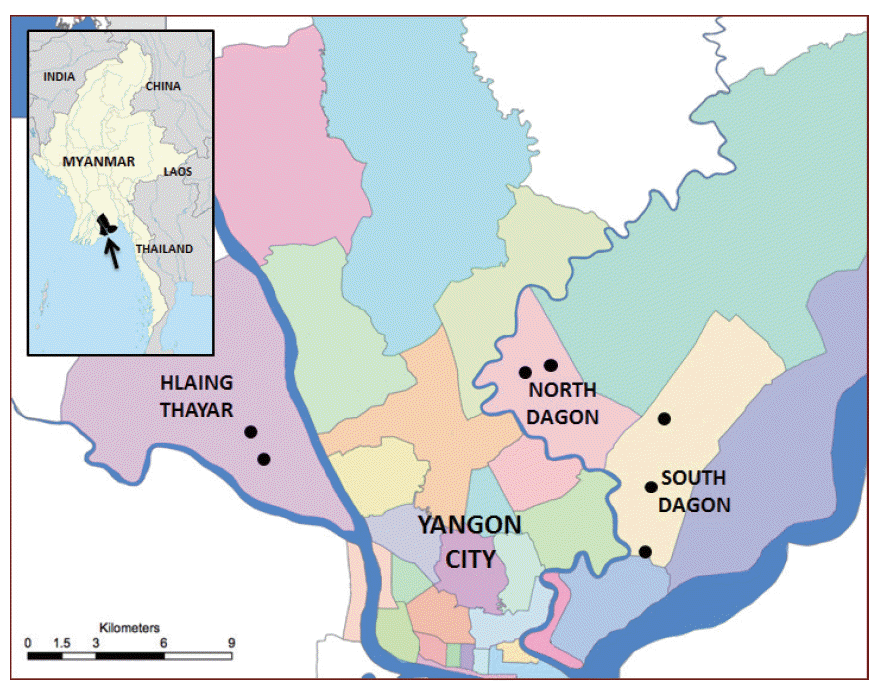
The survey target included 7 primary schools (
Fig. 1) in 3 suburban areas (South Dagon, Hlaing Thayar, and North Dagon) of Yangon. The subjects were 761 primary schoolchildren (383 boys and 378 girls) who were 5-7 years of age. The cellophane tape (cello-tape) anal swab method was used to detect the eggs. The sticky side of the transparent cello-tape was stuck to each child’s perianal area and removed. Collected samples were transported to the National Health Laboratory, Yangon, Myanmar and examined under light microscopy by at least 2 medical specialists. The prevalence of
E. vermicularis eggs was compared between sex and township, and the results were statistically evaluated by the chi-square test.
For identification of the pinworm species, PCR and nucleotide sequencing were performed on the 5S rDNA region according to the procedures reported previously [
6]. Total genomic DNA was extracted from the eggs on the cello-tape by using a DNeasy Blood and Tissue Kit (QIAGEN, Hilden, Germany) with minor modifications. Briefly, the eggs were mechanically detached from the cello-tape using forceps under a stereomicroscope, and the isolated eggs were vortexed with glass beads for lysis. The next step followed the manufacturer’s instructions. The PCR product was amplified by using the Cosmo Labopass X2 PCR Premix kit (Cosmo Genetech, Seoul, Korea) with primers of 5S rDNA (forward primer 5'-CACTTGCTATACCAACAACAC-3'; reverse primer 5'-GCGCTACTAAACCATAGAG-3'), and automated DNA sequencing was performed by Solgent Co. (Daejeon, Korea). Nucleotide sequences obtained were aligned by using the Geneious Program, version 7.1.7 (Geneious Co., Wellington, New Zealand) [
6].
The P-values of <0.05 were considered statistically significant. This study was approved by the National Health Laboratory, Yangon, Myanmar and the Korea Association of Health Promotion, Korea under the agreement of Korea-Myanmar International Project on Intestinal Parasite Control in Primary Schoolchildren around Yangon, Myanmar (2013-2015).
Out of the total 761 primary schoolchildren examined, 359 (47.2%) were positive for the eggs of
E. vermicularis (
Table 1). The positive rate was not significantly (
P >0.05) different between boys (186/383, 48.6%) and girls (173/378, 45.8%). As the age of the examined schoolchildren were the same among most subjects, 5-7 years old, no age-specific different in the prevalence could be noted. However, the prevalence was significantly higher (
P <0.05) in South Dagon (54.6%) than in Hlaing Thayar (43.8%) and North Dagon (34.8%) townships. The prevalence was the highest in boys from South Dagon (56.8%) and the lowest in boys and girls from North Dagon. Sequencing of the 5S rDNA gene of the eggs on the anal swabs showed 99.7% identity (identical in 341 among 342 sites) with
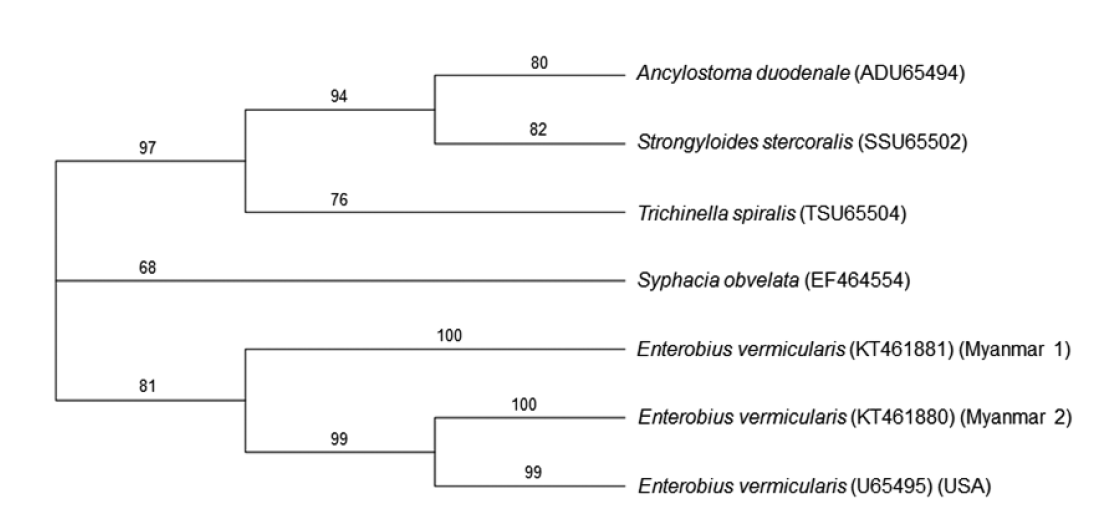
E. vermicularis reported from USA (GenBank no. U65495). The neighbor-joining tree revealed that our 2 specimens (Myanmar 1 & 2, GenBank no. KT461881 and KT461880, respectively), were phylogenetically compatible to
E. vermicularis (
Fig. 2).
The parasite infection rate in specific population such as children and rural residents is particularly attributed to environmental and personal hygiene and sanitation [
7,
8]. For this reason, the primary schoolchildren living in South Dagon seem to live in a poorer socioeconomic status and worse environment than those of the other 2 areas with a higher risk for
E. vermicularis infection. It is locally well known that the general household income and living standards of people in South Dagon is lower than those in Hlaing Thayar and North Dagon townships. Further studies regarding the relationships between the pinworm prevalence and the living standard of people around the surveyed areas in Myanmar should be studied in the near future. By this study, however, it can be concluded that all 3 surveyed areas around Yangon, Myanmar are highly endemic areas of enterobiasis among primary schoolchildren.
There is little information on the general status of intestinal helminth infections in Myanmar, and most surveys were small scale or local hospital-based [
9]. Especially, no studies have been performed on the prevalence of
E. vermicularis infection in Myanmar. In the present study, the egg positive rate (47.2%) of
E. vermicularis was much higher than that reported in other countries; 6.0% [
2], 10.5% [
8], or 18.5% [
3] in South Korea, 0.5% in Taiwan [
10], and 12.1% in Turkey [
11]. However, in a recent report from China, the egg positive rate of children in Guangdong area was as high as 54.9% [
4], slightly higher than that observed in Myanmar in the present study. Interestingly, the egg positive rate of immigrant children from Myanmar who lives in Thailand was 25.2%, a much lower figure than that observed in the present study [
5]. It can be speculated that the immigrant Burmese children residing in Thailand have a better socio-economic status and better environment than in their home villages in Myanmar.
There are 2 main points for effective management and control of
E. vermicularis infection. First, medications should be in mass-treatment style and should be repeated in 2-3 weeks [
12,
13]. The pinworms are easily reinfected, and medication does not kill all pinworm larvae or juvenile worms in the intestine [
12]. Therefore, repeated dose is an important strategy to treat larval/juvenile infections which remained in the host intestine after 1 time medication. Second, Gungoren et al. [
14] reported the effects of hygiene promotion on the risk of reinfection. According to their results [
14], the hygiene promotion group had a much lower risk of reinfection than the treatment only group (no hygiene promotion) or control group (no hygiene promotion, no medication).
The reason for a molecular analysis on the eggs in our study was to rule out a possibility for the present pinworms to be a species different from
E. vermicularis, for example, Enterobius gregorii Hugot, 1983 [
15] and
Syphacia obvelata (Rudolphi, 1802) Seurat, 1916 [
16].
E. gregorii is a human-infecting pinworm species reported to be unique from
E. vermicularis in the morphology of the spicule in male worms [
15]. However,
E. gregorii is considered a synonym of
E. vermicularis by several other workers [
17,
18].
S. obvelata is a pinworm species infecting rodents but can also infect humans [
16]. Our specimens showed a 99.7% homology in the 5S rDNA gene sequence with that of
E. vermicularis in GenBank but distinct from the sequence of
S. obvelata. The 5S rDNA gene sequence of
E. gregorii is unavailable in GenBank.
Taken together, the results of the present study suggest strongly that there is an urgent need to control E. vermicularis infection among schoolchildren in the surveyed areas of suburban Yangon, Myanmar.
Notes
-
The authors declare that they have no conflict of interest related to this study.
We are grateful to the staff of the subjected primary schools in South Dagon, Hlaing Thayar, and North Dagon, Myanmar for their assistance in guidance of the schoolchildren and preparing anal swabs. We also thank the staff of National Health Laboratory, Myanmar and the staff of Korea Association of Health Promotion, Korea who helped this survey. This study was supported by a grant from Seoul National University Hospital (2015).
Fig. 1.Map showing the surveyed areas around Yangon City, Myanmar. The schoolchildren of 7 primary schools (black dots) in 3 communes (South Dagon, Hlaing Thayar, and North Dagon) were subjected in this study.

Fig. 2.A phylogenetic tree based on 5S rDNA gene sequences exploring the relationships of our specimens (Myanmar 1 & 2) with ever-reported sequences in GenBank. Numbers above the branches are bootstrap values. The phylogenetic tree reveals that the 5S rDNA gene sequence from the eggs on cello-tape anal swabs of 2 schoolchildren from Myanmar is compatible with that of E. vermicularis (USA).
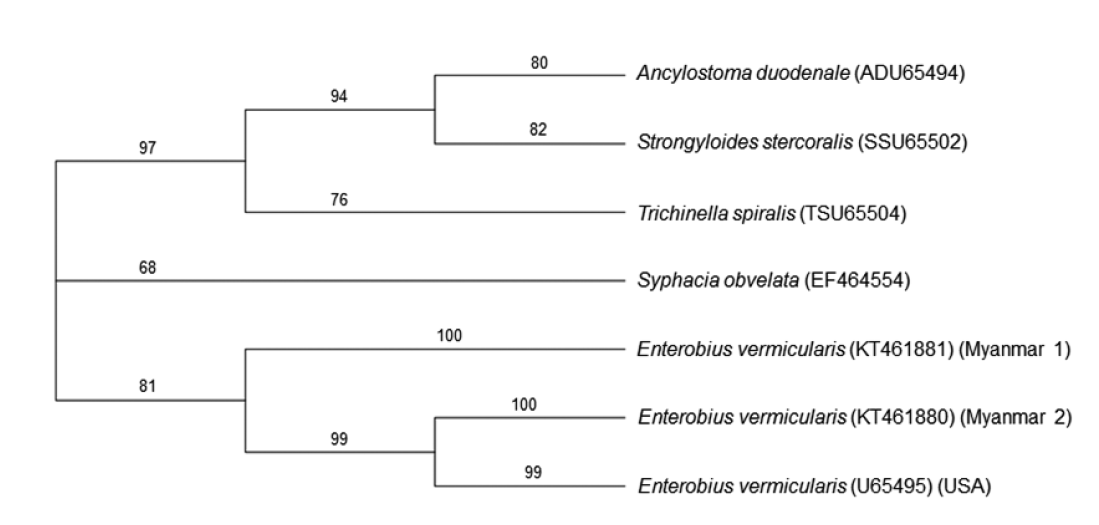
Table 1.The egg positive rate of E. vermicularis among primary schoolchildren in Yangon, Myanmar by cello-tape anal swab examination in 2014
Table 1.
|
Area |
Male
|
Female
|
Total
|
|
No. of exam. |
No. of positive |
(%) |
No. of exam. |
No. of positive |
(%) |
No. of exam. |
No. of positive |
(%) |
|
South Dagon |
199 |
113 |
56.8 |
193 |
101 |
52.3 |
392 |
214 |
54.6a
|
|
Hlaing Thayar |
92 |
41 |
44.6 |
93 |
40 |
43 |
185 |
81 |
43.8a
|
|
North Dagon |
92 |
32 |
34.8 |
92 |
32 |
34.8 |
184 |
64 |
34.8a
|
|
Total |
383 |
186 |
48.6 |
378 |
173 |
45.8 |
761 |
359 |
47.2 |
References
- 1. Bethony J, Brooker S, Albonico M, Geiger SM, Loukas A, Diemert D, Hotez PJ. Soil-transmitted helminth infections: ascariasis, trichuriasis, and hookworm. Lancet 2006;367:1521-1532.
- 2. Kim DH, Cho MK, Park MK, Kang SA, Kim BY, Park SK, Yu HS. Environmental factors related to enterobiasis in a southeast region of Korea. Korean J Parasitol 2013;51:139-142.
- 3. Park JH, Han ET, Kim WH, Shin EH, Guk SM, Kim JL, Chai JY. A survey of Enterobius vermicularis infection among children on western and southern coastal islands of the Republic of Korea. Korean J Parasitol 2005;43:129-134.
- 4. Li HM, Zhou CH, Li ZS, Deng ZH, Ruan CW, Zhang QM, Zhu TJ, Xu LQ, Chen YD. Risk factors for Enterobius vermicularis infection in children in Gaozhou, Guangdong, China. Infect Dis Poverty 2015;4:28.
- 5. Sagnuankiat S, Wanichsuwan M, Bhunnachet E, Jungarat N, Panraksa K, Komalamisra C, Maipanich W, Yoonuan T, Pubampen S, Adisakwattana P, Watthanakulpanich D. Health status of immigrant children and environmental survey of child daycare centers in Samut Sakhon Province, Thailand. J Immigrant Minority Health 2014;1-7. (e-pub ahead of print).
- 6. Lim H, Jung BK, Cho J, Yooyen T, Shin EH, Chai JY. Molecular diagnosis of cause of anisakiasis in humans, South Korea. Emerg Infect Dis 2015;21:342-344.
- 7. Warunee N, Choomanee L, Sataporn P, Rapeeporn Y, Nuttapong W, Sompong S, Thongdee S, Bang-On S, Rachada K. Intestinal parasitic infections among school children in Thailand. Trop Biomed 2007;24:83-88.
- 8. Lee SE, Lee JH, Ju JW, Lee WJ, Cho SH. Prevalence of Enterobius vermicularis among preschool children in Gimhae-si, Gyeongsangnam-do, Korea. Korean J Parasitol 2011;49:183-185.
- 9. Montresor A, Zin TT, Padmasiri E, Allen H, Savioli L. Soil-transmitted helminthiasis in Myanmar and approximate costs for countrywide control. Trop Med Int Health 2004;9:1012-1015.
- 10. Chu TB, Liao CW, Nara T, Huang YC, Chou CM, Liu YH, Fan CK. Enterobius vermicularis infection is well controlled among schoolchildren in nurseries of Taipei City, Taiwan. Rev Soc Brasil Med Trop 2012;45:646-648.
- 11. Alver O, Özakin C, Töre O. The distribution of intestinal parasites detected in the Uludag University Medical faculty. Turkiye Parazitol Derg 2012;36:17-22. (in Turkish).
- 12. Hong ST, Chai JY, Cho SY, Seo BS, Yun CK. Efficacy of repeated chemotherapy in control of Enterobius vermicularis infection. Seoul J Med 1979;20:163-168. (in Korean).
- 13. St Georgiev V. Chemotherapy of enterobiasis (oxyuriasis). Expert Opin Pharmacother 2001;2:267-275.
- 14. Gungoren B, Latipov R, Regallet G, Musabaev E. Effect of hygiene promotion on the risk of reinfection rate of intestinal parasites in children in rural Uzbekistan. Trans R Soc Trop Med Hyg 2007;101:564-569.
- 15. Ahn YK, Chung PR, Soh CT. Enterobius gregorii Hugot, 1983 recovered from school children in Kangwon-do, Korea. Korean J Parasitol 1992;30:163-167. (in Korean).
- 16. Riley WA. A mouse oxyurid, Syphacia obvelata, as a parasite of man. J Parasitol 1919;6:89-93.
- 17. Hasegawa H, Takao Y, Nakao M, Fukuma T, Tsuruta O, Ide K. Is Enterobius gregorii Hugot, 1983 (Namatoda: Oxyuridae) a distinct species? J Parasitol 1998;84:131-134.
- 18. Nakano T, Okamoto M, Ikeda Y, Hasegawa H. Mitochondrial cytochrome c oxidase subunit 1 gene and nuclear rDNA regions of Enterobius vermicularis parasitic in captive chimpanzees with special reference to its relationship with pinworms in humans. Parasitol Res 2006;100:51-57.