Abstract
Malaria transmission remains high in rural Tanzania despite widespread use of insecticide-treated nets and indoor residual spraying. Outdoor biting and flexible host-feeding behavior of Anopheles mosquitoes reduce the effectiveness of these interventions. Livestock near households may influence vector behavior, but evidence from high-transmission rural settings is limited. A cross-sectional study was conducted from November 2022 to February 2023 in 5 villages of Misungwi District, involving 44 households (22 livestock-keeping and 22 non-livestock-keeping). Adult Anopheles mosquitoes were collected indoors and outdoors using Center for Disease Control light traps, identified morphologically and by PCR, and blood-meal sources were determined by ELISA. Host-feeding patterns were assessed using the human blood index, bovine blood index, and foraging ratios. A total of 611 female mosquitoes were collected, dominated by An. gambiae s.l. (96.1%) and An. funestus s.l. (3.9%). Livestock households had more mosquitoes (61.5%) and higher outdoor activity (67.3%), while non-livestock households had higher indoor collections (73.6%). Among 231 blood-fed mosquitoes, 150 (64.9%) were from livestock households and 81 (35.1%) from non-livestock households. In livestock households, 108 (72.0%) had animal blood only, 27 (18.0%) mixed blood, and 15 (10.0%) human blood only. In non-livestock households, 44 (54.3%) had human blood only, 24 (29.6%) mixed blood, and 13 (16.0%) animal blood only. An. arabiensis was opportunistic, while An. gambiae s.s. and An. funestus s.s. remained strongly anthropophilic. Livestock shifts feeding toward animals and increases outdoor activity but does not eliminate human feeding, highlighting the need for integrated malaria control strategies.
-
Key words: Malaria vectors, Anopheles, livestock, blood meal, Tanzania
Introduction
Malaria remains a major global health challenge, with an estimated 263 million cases and nearly 597,000 deaths in 2023, most occurring in sub-Saharan Africa [
1]. Tanzania remains highly endemic, with 93% of the population at risk and 3.6 million cases reported in 2023 [
1]. Despite widespread use of insecticide-treated nets (ITNs) and indoor residual spraying (IRS), residual transmission persists [
2,
3], driven in part by behavioral adaptations in vectors such as the
Anopheles gambiae complex and
An. funestus, which increasingly feed outdoors, bite earlier, and exhibit flexible host preferences [
4,
5].
Livestock keeping is common in rural Tanzania, where more than 60% of households own domestic animals [
6]. In the Lake Zone, particularly Mwanza Region, cattle, goats, sheep, dogs and poultry are often kept within or near household compounds [
7,
8]. In Misungwi District, livestock corrals are frequently located within 20 meters of human dwellings [
8]. This close proximity increases opportunities for human–animal–vector interactions, yet limited empirical evidence exists on how livestock ownership influences mosquito host choice and feeding behavior in such settings.
Studies across sub-Saharan Africa suggest that livestock may divert mosquito feeding away from humans (zooprophylaxis) or, conversely, sustain vector populations by providing alternative blood sources (zoopotentiation) [
8,
9]. These effects vary widely across ecological contexts, and it remains unclear how livestock presence interacts with household factors, vector species composition, and vector control measures to shape mosquito feeding patterns.
Blood meal analysis provides a direct method to assess host use and can reveal species-specific feeding behavior and associations with household characteristics [
10-
12]. This study examined blood meals of female
Anopheles mosquitoes collected from livestock-keeping and non-livestock households in Misungwi District, northwestern Tanzania. Specifically, we aimed to (i) quantify human, animal, and mixed blood meals; (ii) compare indoor and outdoor feeding patterns relative to livestock presence; and (iii) assess how vector species identity and abundance relate to host choice. Findings will support improved malaria control strategies in livestock-keeping communities and similar ecological settings.
Methods
Ethics statement
The protocols for this study were approved by the National Institute for Medical Research (NIMR), a department of the Ministry of Health in Tanzania (NIMR/HQ/R.8a/Vol. IX/4114). All household owners were consulted and consented prior to their involvement in the study. Center for Disease Control (CDC) traps were placed indoors or outdoors in only those households that consented to participate.
Study area
The study was conducted in Misungwi District, Mwanza Region, northwestern Tanzania (
Fig. 1). Geographically, the district lies at approximately 2°51′59″ S and 33°05′60″ E, covering an area of 2,553 km² and hosting a population of 467,867 people [
13]. Misungwi is largely rural, with most residents engaged in mixed farming and livestock keeping. The district hosts a substantial livestock population, including cattle, goats, sheep, poultry, and dogs. Misungwi and Mabuki wards were purposively selected due to their high malaria transmission and close proximity of livestock to human dwellings, characteristics that provide suitable conditions for investigating vector feeding behavior (
Fig. 1B). Malaria transmission remains moderate to high, peaking after the 2 rainy seasons, with a reported prevalence of approximately 52% across all age groups [
14]. ITNs are the primary malaria control tool, while IRS has not been conducted since 2015. The coexistence of persistent malaria transmission, high vector densities, and widespread livestock ownership makes Misungwi an important site for examining vector–host interactions.
A total of 44 households from 5 villages were selected for mosquito collection: 3 villages from Misungwi ward (Old Misungwi, Mwamanga, and Iteja) and 2 from Mabuki ward (Lubuga and Mwanangwa) (
Fig. 1C). The villages were purposively selected based on previous reports identifying crop cultivation and pastoralism as the primary economic activities in these areas. Households were randomly selected from lists provided by district authorities. Among the selected households, 22 kept livestock and 22 did not. Using a structured questionnaire and field measurements, the number of vertebrate hosts and the distances between livestock corrals and human dwellings were recorded. In the study setting, livestock enclosures are typically constructed in close proximity to sleeping structures due to cultural husbandry practices, land-use constraints, and security considerations, resulting in natural spatial clustering within short peri-domestic distances. Field measurements indicated that most corrals were located within approximately 10–15 meters of human dwellings. Accordingly, the distance between corrals and human dwellings was categorized into 2 groups: 1–12 meters and ≥13 meters. Corrals located within 1–12 meters were classified as “close,” while those ≥13 meters were classified as “distant.” This classification was informed by both the observed settlement structure and biologically plausible attraction ranges of host-seeking mosquitoes. Mosquito host-seeking behavior in peri-domestic environments typically operates at short spatial scales, with attraction gradients frequently documented within 10–20 meters of host sources [
15,
16]. Similar short-range thresholds have been used in studies examining livestock proximity, zooprophylaxis, and household-level malaria exposure [
17].
Mosquito collections were conducted over a 4-month period, from November 2022 to February 2023. In the selected households, adult mosquitoes were sampled both indoors and outdoors using CDC miniature light traps. The traps were set in the evening and operated between 18:00 and 06:00 hours, following standard protocols for malaria surveillance [
18]. Each household had 2 CDC light traps set per night, one indoors and one outdoors. The indoor trap was suspended about 1.5 meters above the floor at the foot end of a bed in a sleeping area, as described previously [
19]. For outdoor collections in livestock-keeping households, the trap was positioned within the livestock corral at approximately 1.5 meters above ground level and at least 1 meter away from large animals to prevent physical obstruction, while remaining within the host odor plume zone. In households without livestock, the outdoor trap was placed within 10 meters of the main house at approximately 1.5 meters above ground level in an open peri-domestic area. To ensure comparability across households, traps were positioned in similar heights across all selected houses, away from artificial light sources, dense vegetation, cooking areas, and obvious mosquito resting sites, and were placed in locations with similar exposure to human activity and airflow. Placement criteria were standardized across households to minimize environmental heterogeneity and reduce sampling bias related to microhabitat differences [
20].
Collected mosquitoes were morphologically identified to distinguish
Anopheles species from other mosquito genera. Identification was based on standard African vector identification keys including body colouration, sex, presence of pale or dark scales on the palps and proboscis, wing scale patterns (notably black scales), and the structure of the terminal abdominal segments [
21,
22]. All adult female
Anopheles were preserved individually in silica gel and transported to the NIMR Mwanza Centre for molecular confirmation and blood meal analysis. At the NIMR laboratory, each mosquito was examined under a dissecting microscope, and dissection was performed by transversely separating the thorax from the abdomen. The legs were preserved for species-specific molecular identification, and the abdomens of blood-fed mosquitoes were used for blood meal source analysis. For downstream analyses, including blood meal source identification, host preference, and foraging ratio (FR) calculations, mosquito species identity was assigned based on molecular confirmation whenever available. Specimens that failed PCR amplification were retained under their morphologically identified group. This approach allowed maximal inclusion of specimens while maintaining species-level resolution [
23].
For species confirmation within the
An. gambiae and
An. funestus complexes, PCR targeting the ITS2 region was conducted using species-specific primers following established protocols [
17,
22]. Briefly, genomic DNA was mixed with the following primers in a 20 µl reaction: a universal forward primer common for all species (UN: 5’-GTG TGC CCC TTC CTC GAT GT-3’) and species-specific reverse primers for
An. gambiae s.s. (GA: 5’-CTG GTT TGG TCG GCA CGT TT-3’),
An. arabiensis (AR: 5’-AAG TGT CCT TCT CCA TCC TA-3’),
An. funestus (FUN: CTC GGG CAT CGA TGG GTT AAT CATG), and (PAR: GCC CTG CGG TCC CAA GCT AGA TT) for
An. parensis. The selection of primers was based on previous entomological surveys [
24,
25]. Reactions were carried out in a 20 µl volume using AccuPower PCR PreMix (Bioneer). A 1 µl of extracted gDNA was used as a template for the PCR reaction, mixed with 25 pmol of primers GA, AR, FUN and PAR and 0.25 pmol of UN (Eurogentec). The PCR conditions were as follows: pre-denaturation at 94°C for 4 min, followed by 30 cycles of denaturation for 30 sec at 94°C, annealing for 30 sec at 58°C, and extension for 45 sec at 72°C, plus a final extension at 72°C for 7 min. Amplified PCR products were visualized on 2% agarose gels, stained with ethidium bromide. A known
An. arabiensis strain from the Sekoru colony (Jimma, Ethiopia), was used as a positive control and double-distilled water used as negative control. These molecular analyses provided confirmatory evidence supporting the morphological identification.
To identify the vertebrate host sources of blood-fed
Anopheles mosquitoes collected both indoors and outdoors, ELISA was performed on the abdomens of morphologically and molecular identified specimens using established methods [
26]. The analysis utilized species-specific antibodies targeting human, bovine, caprine, and canine antigens, corresponding to the most commonly kept domestic animals in the study area [
12]. Briefly, each dissected mosquito abdomen was homogenized in 50 µl of grinding buffer and applied to ELISA plates pre-coated with capture monoclonal antibodies. Plates were then blocked with 200 µl of blocking buffer and incubated at room temperature for 1 h. Following this, homogenized samples, along with positive and double-distilled water as negative controls, were added to the wells and incubated for 2 h. After incubation, the plates were washed twice, and 50 µl of peroxidase-conjugated monoclonal antibody was added to each well, followed by a 1-h incubation in the dark. Plates were then washed 3 times with phosphate-buffered saline containing 0.05% Tween-20, and 100 µl of substrate solution was added to each well. After a final 30-min incubation in the dark, absorbance was measured at 405 nm using an ELISA plate reader. Samples with optical density values exceeding the negative control threshold were classified as positive for the corresponding host. This method provided reliable confirmation of mosquito host-feeding preferences and supported further analysis of vector–host interactions within the ecological and household contexts of the study area.
Data were entered and cleaned in MS Excel file (Microsoft), then analyzed using GraphPad Prism version 8.0.2 (GraphPad Software). Prior to statistical comparisons, data on blood meal sources (host types), human blood index (HBI), and bovine blood index (BBI) were tested for normality and log-transformed. HBI and BBI were calculated as the proportions of mosquitoes with human and bovine blood meals, respectively, among the total blood-fed specimens identified. Mixed (human + bovine) meals were included in both HBI and BBI calculations. Differences in HBI and BBI between indoor and outdoor collections were assessed using independent
t-tests (
P<0.05). Variation in mosquito host choices was analyzed using one-way ANOVA, followed by Tukey’s HSD test for post hoc comparisons (
P<0.05). FRs were calculated to assess host selection by mosquitoes [
27], using the formula: FR=(NAE/NTE)/(NAP/NTP), where NAE is the number of engorged mosquitoes feeding on a specific host, NTE is the total engorged mosquitoes, NAP is the number of that host in the area, and NTP is the total number of all host types [
27]. An FR ≈1 indicates opportunistic feeding; FR >1 suggests preference, and FR <1 indicates avoidance [
12,
28]. FRs were computed for humans, cattle, goat, sheep, chicken and dogs. Host feeding indices (HFIs) were also calculated to compare feeding preferences between host pairs using the formula: HFI=(Nx/Ny)/(Ax/Ay), where Nx and Ny are the mean number of mosquitoes feeding on hosts x and y, and Ax and Ay are the corresponding host abundances. An HFI of 1 indicates no preference; >1 suggests preference for host x; <1 suggests preference for host y. In this study, HFI was calculated specifically for humans versus cattle.
Results
Collection, identification and distribution of Anopheles mosquitoes
Morphological identification confirmed 611 female
Anopheles mosquitoes. PCR successfully amplified 553 specimens (90.5%), while 58 (9.5%) did not amplify (
Table 1). Of the 587 mosquitoes morphologically identified as
An. gambiae s.l., 535 (91%) amplified, revealing 446 (75.9%)
An. arabiensis and 89 (15.1%)
An. gambiae s.s., with 52 (9%) unamplified. Among 24 mosquitoes identified morphologically as
An. funestus s.l., 18 (75%) amplified and were all confirmed as
An. funestus s.s. Overall, 376 mosquitoes (61.5%) were collected from livestock-keeping households and 235 (38.5%) from non-livestock households (
P=0.5445), indicating no evidence of a difference in overall mosquito abundance between household types. In livestock-keeping households, more mosquitoes were collected outdoors (253, 67.3%) than indoors (123, 32.7%); however, this difference was not statistically significant (
P>0.05). In contrast, non-livestock households had significantly higher indoor collections (173, 73.6%) than outdoor (62, 26.4%) (
P=0.0361). Spatial distribution across villages did not differ significantly (
P>0.05), indicating no detectable variation in mosquito abundance between villages during the study period (
Table 2).
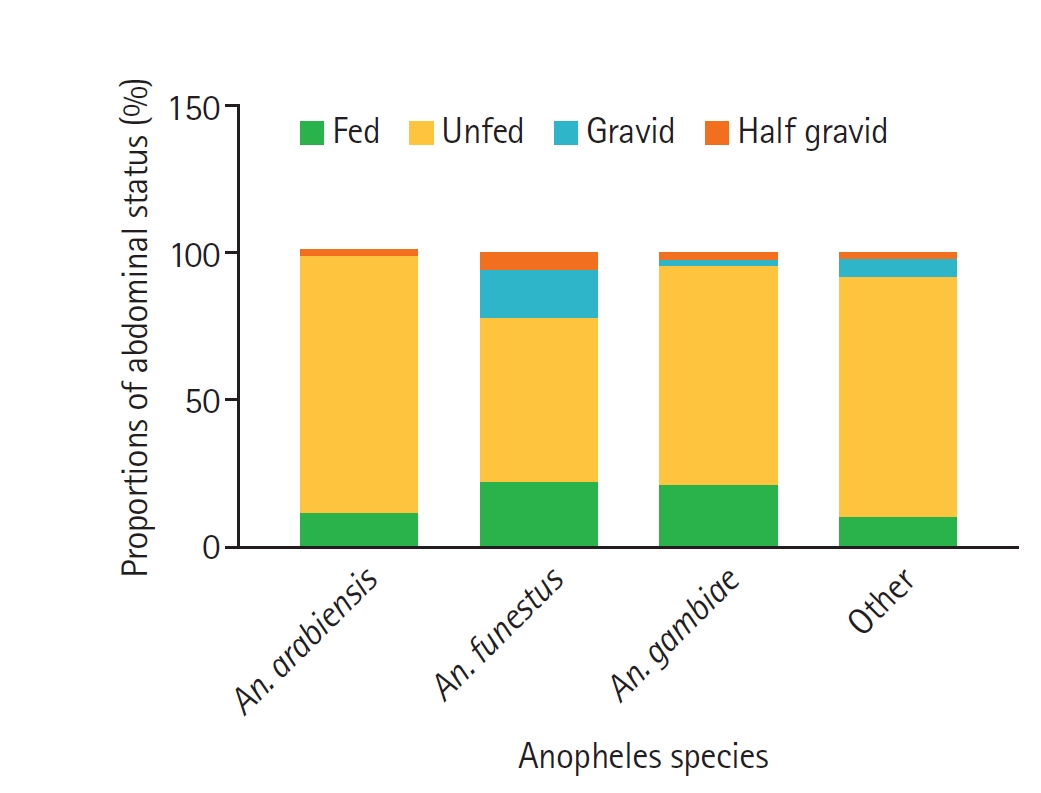
The abdominal status of the 611 adult female
Anopheles mosquitoes collected is summarized in
Table 3 and
Fig. 2. The majority of the collected
Anopheles were unfed (362, 59.2%), followed by fed (231, 37.8%), gravid (11, 1.8%), and half-gravid (7, 1.1%). A higher proportion of fed (150, 64.9%), unfed (217, 59.9%) and gravid (7, 63.6%) female
Anopheles mosquitoes were collected from livestock-keeping households compared to non-livestock-keeping households that had 81 (35.1%), 145 (40.1%), and 4 (36.4%), respectively. In the livestock-keeping households, the fed, unfed and gravid
Anopheles mosquitoes were significantly found outdoors, 229 (60.9%), than indoors, 147 (39.1%) (
P<0.05). This was contrary to the observation in non-livestock-keeping households where a large proportion of
Anopheles mosquitoes were collected from indoors 183 (77.9%) than outdoors 52 (22.1%) (
P<0.05).
A total of 12,388 hosts were recorded across the 44 surveyed houses in 5 villages, of which 6,524 (52.7%) were from Misungwi ward and the remainder from Mabuki ward. Of the 3 villages from Misungwi ward, Old Misungwi accounted for 4,324 (34.9%) followed by Iteja 1,295 (10.5%) and Mwamanga 905 (7.3%). From Mabuki ward, the number of hosts were large in Mwanangwa 3,710 (29.9%) than Lubuga 2,154 (17.4%). The identified hosts included cows 8,226 (66.4%), humans 2,253 (18.2%), sheep (7.9%), chickens (3.1%), goats (2.8%), and dogs 201 (1.6%). A large proportion of the domesticated animals, 6,939/10,135 (68.4%), were kept in corrals located less than 12 meters from human dwellings, compared to 3,196 (31.5%) housed more than 12 meters away (
P=0.0062). Overall, a higher proportion of human and cattle hosts was recorded in the study area (F
5,20=5,927,
P=0.0016) (
Table 2).
As indicated in
Table 3, fed
Anopheles mosquitoes were 231/611 (37.8%) and all were assayed for bloodmeal source analysis using ELISA. Of the assayed mosquitoes, 150 (64.9%) were collected from livestock-keeping houses and 81 (35.1%) from non-livestock-keeping households. A large proportion of the fed
Anopheles mosquitoes were from Mabuki ward (159; 68.8%), which comprised Mwanangwa and Lubuga villages. Of these 159 mosquitoes, 114 (71.7%) from Mwanangwa village and 45 (28.3%) from Lubuga village. The remaining 72 (31.2%) fed
Anopheles mosquitoes were from Misungwi ward, consisting of Old Misungwi (33, 45.8%), Iteja (18, 25.0%), and Mwamanga (21, 29.2%). Although numerical differences were observed in the distribution of fed mosquitoes across villages, these differences were not statistically significant (
P>0.05).
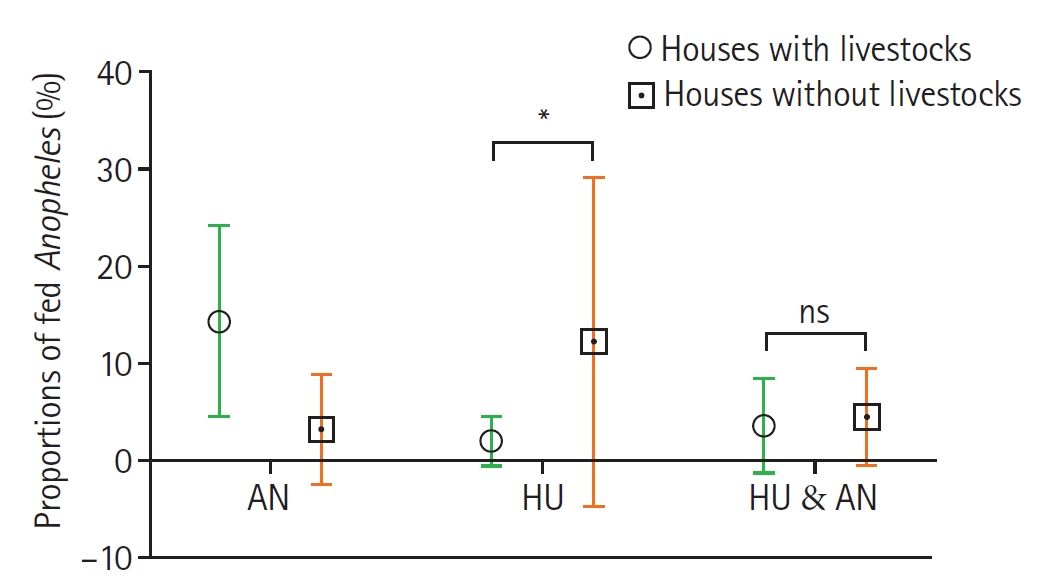
Among the 150 fed
Anopheles collected from livestock-keeping households, 108 (72.0%) contained animal blood, 27 (18.0%) had mixed human and other animal blood, and 15 (10.0%) had human blood. This was contrary to non-livestock keeping households where of the 81-fed
Anopheles, 44 (54.3%) had human blood, 24 (29.6%) had human and other animal blood, and 13 (16.0%) had only animal blood (
Fig. 3). Comparatively, the proportion of mosquitoes fed on human blood was higher in non-livestock keeping houses 68 (29.4%) compared to those from livestock-keeping houses 42 (18.2%) and the difference was significant (
P=0.0200). Likewise, mosquitoes with animal blood only were significantly higher in livestock keeping houses 108 (46.8%) compared to non-livestock keeping houses 13 (5.6%) (
P=0.0317) (
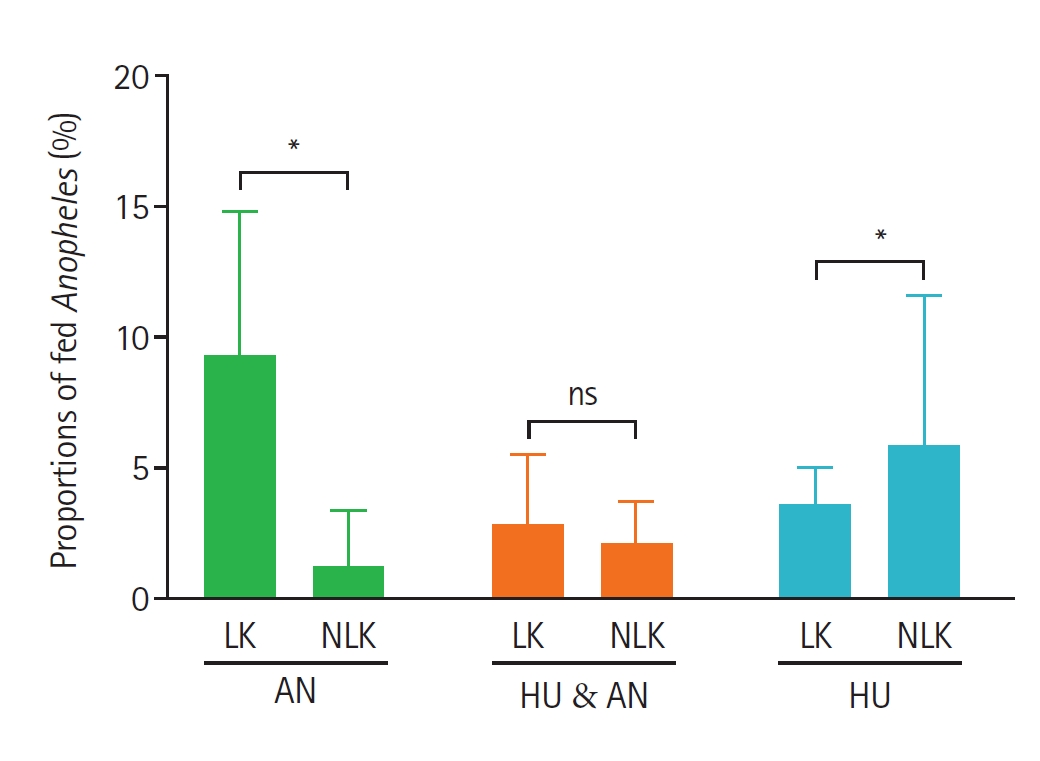
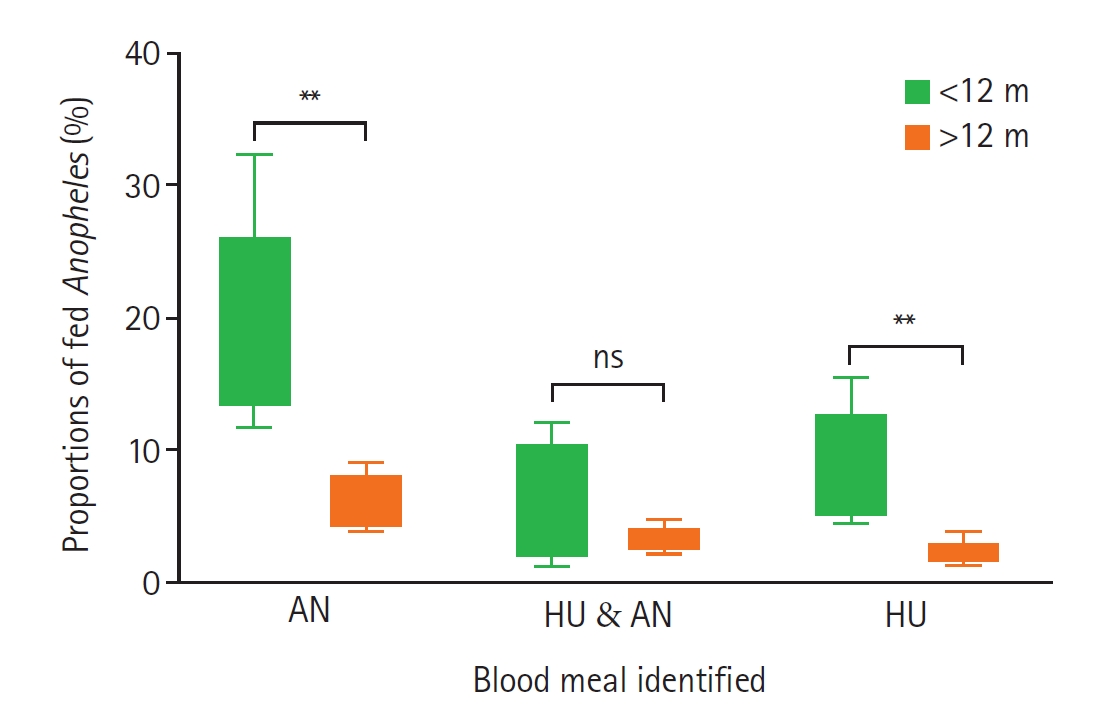
Fig. 4). Mosquitoes collected from corrals situated closer to houses (<12 m) showed a higher proportion of animal, mixed (animal + human), and human blood meals compared to those from corrals located farther away (>12 m) (
Fig. 5).
The FR analysis was calculated for all identified vertebrate hosts (humans, cattle, goats, sheep, chickens, and dogs) to evaluate feeding preference relative to host availability (
Table 4). Distinct host-selection patterns were observed among the 3
Anopheles species.
An. gambiae s.s. and
An. funestus exhibited strong anthropophilic tendencies, with high FR values for humans (FR=4.4 and 4.95, respectively), despite relatively lower human availability. Both species showed consistent avoidance of livestock hosts (FR <1 across cattle, goats, sheep, chickens, and dogs). In contrast,
An. arabiensis demonstrated opportunistic feeding behavior, with a moderate preference for goats (FR=2.86), a slight preference for humans (FR=1.38), and near-random feeding on sheep, chickens, and dogs. Although cattle represented the most abundant host type (66.4%), their FR values remained below 1 across all vector species, suggesting relative avoidance rather than proportional feeding. These findings indicate that host selection was not solely determined by host abundance but reflected species-specific behavioral traits and ecological flexibility (
Table 4).
Discussion
In many African settings, particularly in rural and peri-urban areas where livestock ownership is common, close human–livestock cohabitation provides multiple blood-meal sources that can influence mosquito host-seeking behavior and feeding preferences. Increasing evidence indicates that malaria vectors historically associated with indoor feeding and resting are shifting toward outdoor and animal-associated behaviors, thereby reducing the effectiveness of indoor-based interventions such as ITNs and IRS [
29]. Studies from Ethiopia, Kenya, and Tanzania have consistently shown that
An. arabiensis, a key member of the
An. gambiae complex, exhibits substantial behavioral plasticity, switching between human and animal hosts depending on host availability and environmental conditions [
8,
30,
31]. Understanding the role of livestock in shaping vector behavior is therefore essential for designing effective malaria control strategies in settings where residual transmission persists despite high ITN and IRS coverage.
Our findings indicate that livestock keeping was associated with differences in vector feeding behavior and indoor–outdoor distribution patterns; however, not all observed differences, particularly in overall mosquito abundance and village-level distribution, reached statistical significance.
An. arabiensis was the predominant vector species and was more frequently encountered outdoors in livestock-keeping households, consistent with its documented exophagic and zoophilic tendencies [
32,
33]. In contrast,
An. gambiae s.s. and
An. funestus s.s. remained strongly anthropophilic, reinforcing their continued role in sustaining indoor malaria transmission even in areas with widespread indoor vector control [
34]. Statistically significant differences in indoor–outdoor mosquito densities were observed in non-livestock households, whereas the corresponding pattern in livestock-keeping households did not reach statistical significance. Therefore, while livestock presence may influence host-seeking and resting behavior, this interpretation should be made cautiously and primarily with respect to feeding patterns, which showed clearer statistical support.
Blood-meal analysis further revealed distinct feeding patterns between household types. Mosquitoes collected from livestock-keeping households exhibited a higher proportion of animal or mixed blood meals, whereas those from non-livestock households fed more frequently on humans. The reduced HBI observed in livestock-keeping settings is consistent with a potential zooprophylactic effect, whereby domestic animals divert mosquito feeding away from humans [
27]. However, HBI reflects feeding behavior rather than transmission intensity, and a reduction in HBI alone does not necessarily translate into a measurable reduction in malaria transmission risk. Transmission dynamics depend on multiple entomological and epidemiological parameters, including vector density, survival rates, sporozoite prevalence, entomological inoculation rates, and human infection prevalence. In some settings, livestock-associated blood sources may sustain or even increase vector populations despite reduced anthropophily, thereby maintaining transmission potential [
27,
35]. Because this study did not assess sporozoite rates, entomological inoculation rates, or human malaria prevalence, we cannot directly determine whether the observed behavioral shift results in reduced malaria risk. These findings should therefore be interpreted as evidence of altered vector feeding patterns rather than definitive evidence of a protective zooprophylactic effect. Future studies integrating behavioral, entomological, and epidemiological indicators will be essential to clarify the net impact of livestock ownership on malaria transmission dynamics.
Host preference analysis revealed clear interspecific differences in feeding behavior.
An. gambiae s.s. and
An. funestus s.s. remained highly anthropophilic, with FR values exceeding 4 for humans and consistent avoidance of livestock hosts, reinforcing their established role as primary indoor malaria vectors. In contrast,
An. arabiensis exhibited greater behavioral plasticity, showing a strong preference for goats (FR=2.86), a slight preference for humans (FR=1.38), and largely neutral or random feeding on other domestic animals. Despite the numerical dominance of cattle in the study area, their FR values were consistently below one across species, indicating that feeding patterns were not strictly proportional to host availability. These results underscore the ecological adaptability of
An. arabiensis and its capacity to exploit multiple host types, a trait that may facilitate its persistence in livestock-associated and outdoor environments under sustained indoor vector control pressure [
34].
These findings have important implications for malaria control in Tanzania. The continued presence of highly anthropophilic vectors underscores the ongoing importance of ITNs and IRS for reducing indoor transmission. However, the predominance of An. arabiensis, particularly in outdoor collections, highlights a potential behavioral gap that may contribute to residual transmission beyond the reach of indoor interventions. This interpretation is primarily supported by species composition and feeding data rather than differences in overall mosquito abundance, which were not statistically significant. The close proximity of livestock shelters to human dwellings further increases opportunities for mosquito feeding and resting near both hosts. Addressing this challenge will require integrated vector management strategies that complement indoor tools with targeted outdoor and livestock-based approaches, including insecticide-treated livestock, improved corral management, increased separation between animal shelters and human dwellings, larval source management, and other outdoor control measures. Such integrated approaches may enhance the effectiveness of existing interventions and contribute to reducing residual malaria transmission in endemic rural settings.
Study limitations
Despite its strengths, this study has several limitations. The cross-sectional design and 4-month data collection during the rainy season limit assessment of temporal dynamics and seasonal variability in mosquito populations and host–vector interactions, and may not reflect year-round fluctuations in abundance, species composition, and livestock–human contact patterns. The relatively small sample size (n=44 households) may have reduced statistical power, increased the risk of type II error, and limited generalizability. Additionally, categorizing livestock–dwelling distance into 2 groups (1–12 m and ≥13 m), although informed by settlement patterns and biologically plausible mosquito attraction ranges, may oversimplify a potentially continuous and nonlinear relationship with mosquito density. This approach was partly driven by analytical constraints, as further stratification could have produced sparse data and unstable estimates. A small proportion of specimens (≈9.5%) could not be confirmed molecularly, and were retained under their morphological classification, which may have introduced minor misclassification in species-level analyses, including host preference and FRs. Future longitudinal studies across multiple seasons, with larger samples and spatially explicit analyses modeling distance as a continuous variable, are needed to better characterize temporal and distance-dependent vector dynamics and strengthen causal inference and external validity.
Notes
-
Author contributions
Conceptualization: Zakayo D, Matiya DJ, Kidima W, Mazigo E. Data curation: Kidima W, Mazigo E. Formal analysis: Zakayo D, Kidima W, Mazigo E. Funding acquisition: Kwon BI. Investigation: Zakayo D, Matiya DJ, Kidima W, Mazigo E. Methodology: Zakayo D, Matiya DJ, Kidima W, Mazigo E. Project administration: Matiya DJ, Kidima W, Mazigo E. Resources: Mazigo E. Software: Matiya DJ, Mazigo E. Supervision: Matiya DJ, Kidima W, Mazigo E. Validation: Zakayo D, Matiya DJ, Kidima W, Kwon J, Kwon BI, Mazigo E. Visualization: Zakayo D, Matiya DJ, Kidima W, Kwon J, Mazigo E. Writing – original draft: Zakayo D, Kidima W. Writing – review & editing: Matiya DJ, Kidima W, Kwon J, Kwon BI, Mazigo E.
-
Conflict of interest
The authors have no conflicts of interest to declare.
-
Funding
This research was supported by the Regional Innovation System and Education (RISE) program through the Gangwon RISE Center, funded by the Ministry of Education and the Gangwon State, Republic of Korea (2025-RISE-10-005).
-
Acknowledgments
We thank the study communities in Misungwi District for their cooperation and participation. We are grateful to the local leaders for facilitating field activities and to the field and laboratory teams for their technical support during mosquito collection, identification, and laboratory analyses. We also acknowledge the support of the NIMR Mwanza Centre for providing laboratory facilities and logistical assistance throughout the study.
Fig. 1.Study sites. (A) Map of Tanzania showing the location of Mwanza Region and associated study sites. (B) Mwanza Region highlighting Misungwi District Council and the 2 purposively selected wards for the study, Misungwi and Mabuki. (C) Misungwi and Mabuki wards indicating the 5 villages chosen for the study, with 3 villages from Misungwi ward and 2 from Mabuki ward. Villages were purposively selected based on previous reports identifying crop cultivation and pastoralism as the primary economic activities in the areas. The maps were created using QGIS version 3.34.1.
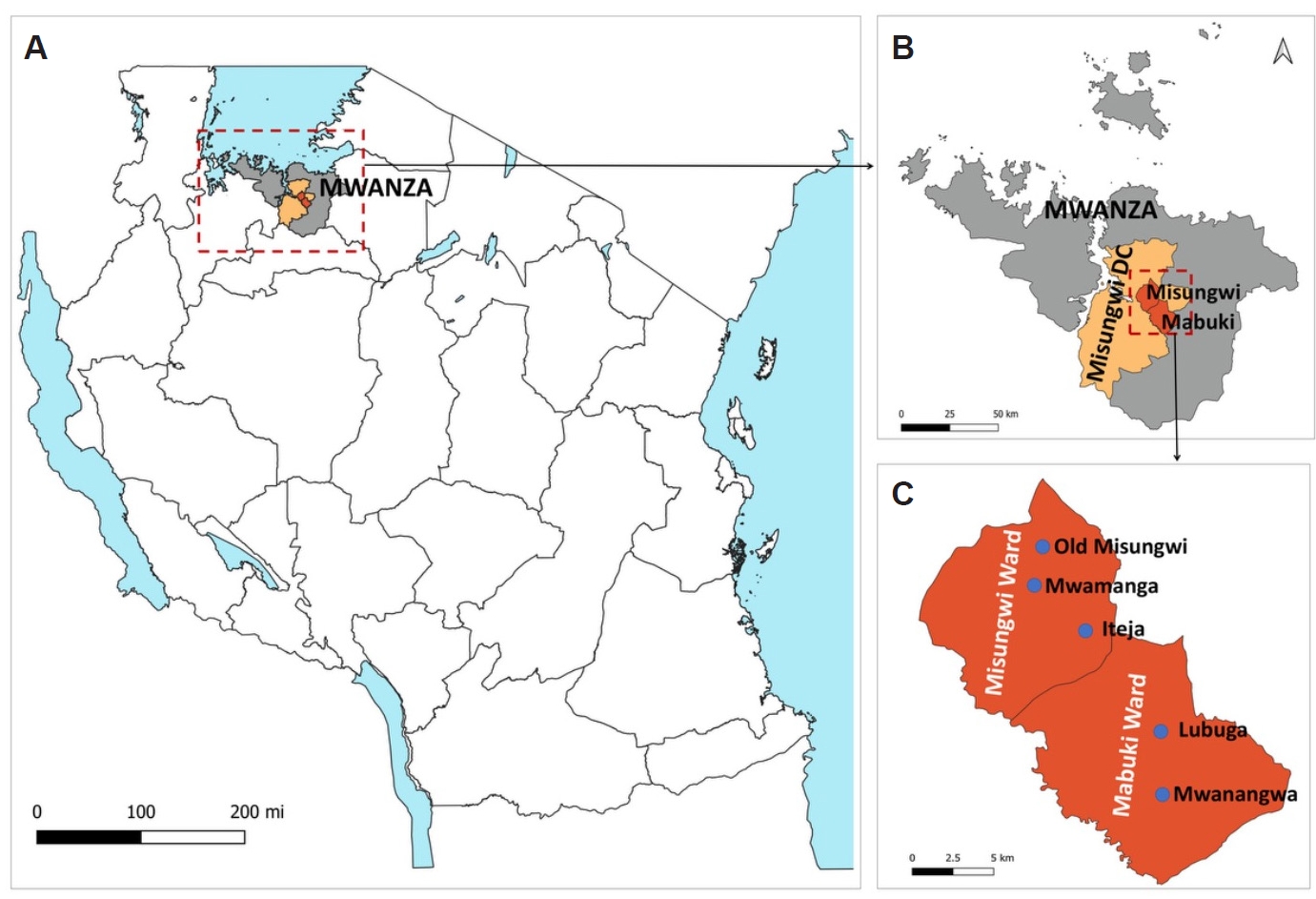
Fig. 2.Proportions of abdominal status of female Anopheles species collected in the study areas. Bars represent the relative proportions of fed, unfed, gravid, and half-gravid females within each Anopheles species group (An. arabiensis, An. funestus, An. gambiae, and other Anopheles species). “Fed” indicates females with freshly ingested blood, “unfed” those without visible blood, “gravid” females with fully developed eggs, and “half-gravid” females with partially developed eggs.
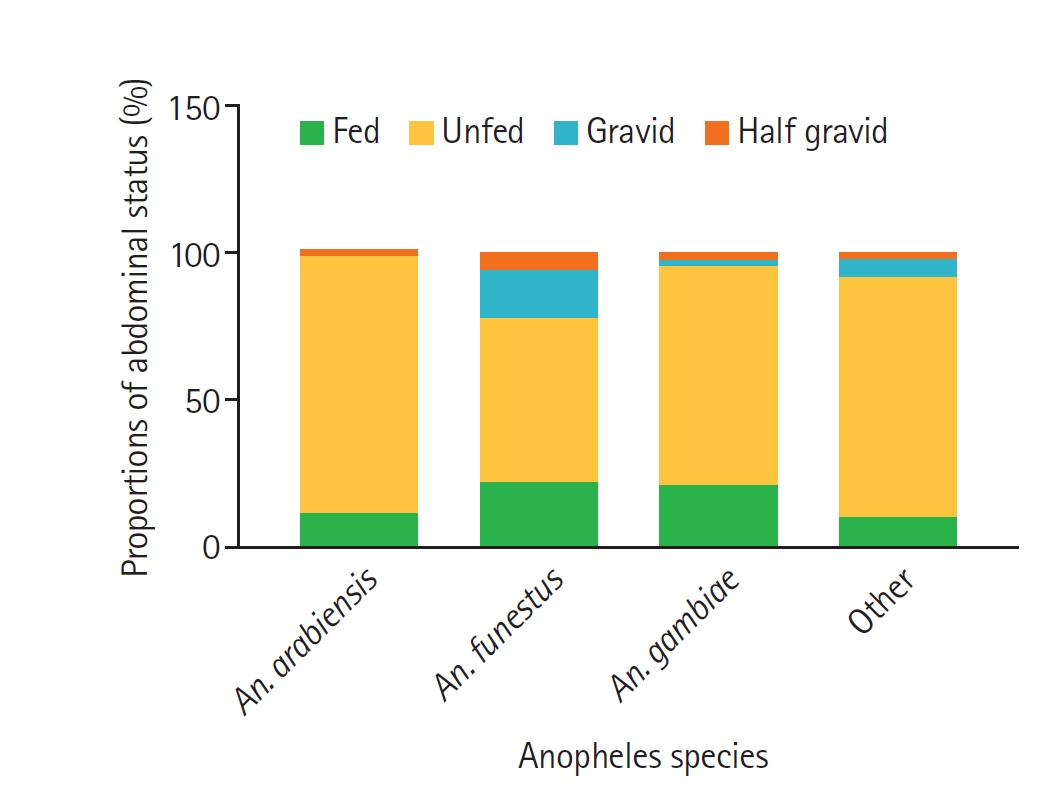
Fig. 3.Proportions of fed Anopheles mosquitoes collected from houses with and without livestock. Bars show the mean±SE proportions of blood-fed Anopheles in different host categories: animal husbandry host (AN), human host (HU), and HU & AN (mixed blood meal index of human and animal hosts). Circles represent livestock-keeping households, while squares represent non–livestock-keeping households. Asterisks indicate statistically significant differences (P<0.05), and “ns” denotes a non-significant difference between the 2 household groups by Mann–Whitney test at 95% confidence interval.
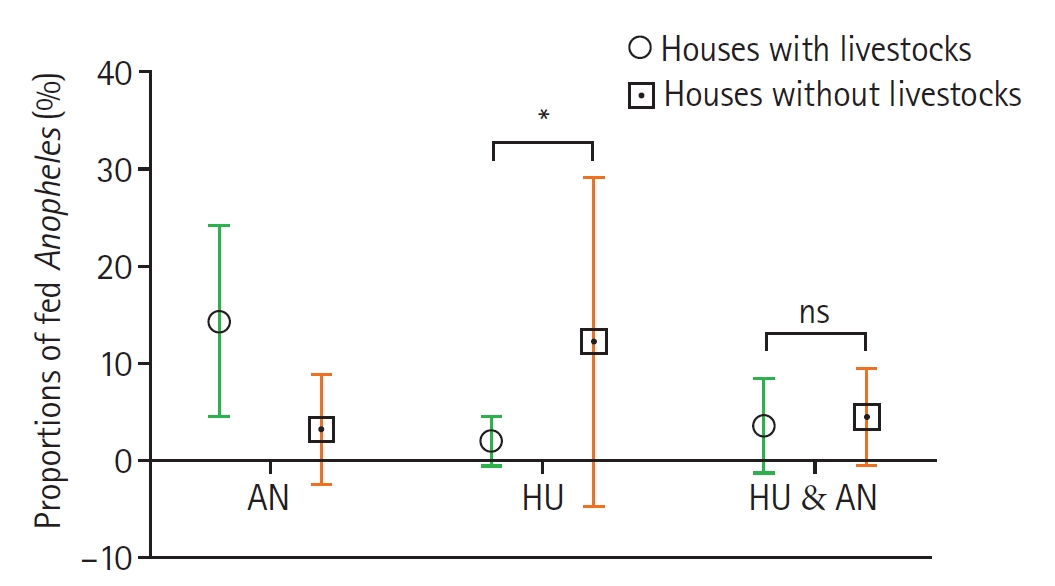
Fig. 4.Proportions of fed Anopheles mosquitoes collected from livestock-keeping (LK) and non-livestock-keeping (NLK) households. Bars represent the mean proportions (±SD) of fed Anopheles associated with different host categories: animal hosts (AN), human and animal hosts (HU & AN), and human hosts (HU). Statistical comparisons between LK and NLK groups were performed using the Mann–Whitney test. Asterisks indicate statistically significant differences (P<0.05). ns, not significant at 95% confidence interval.
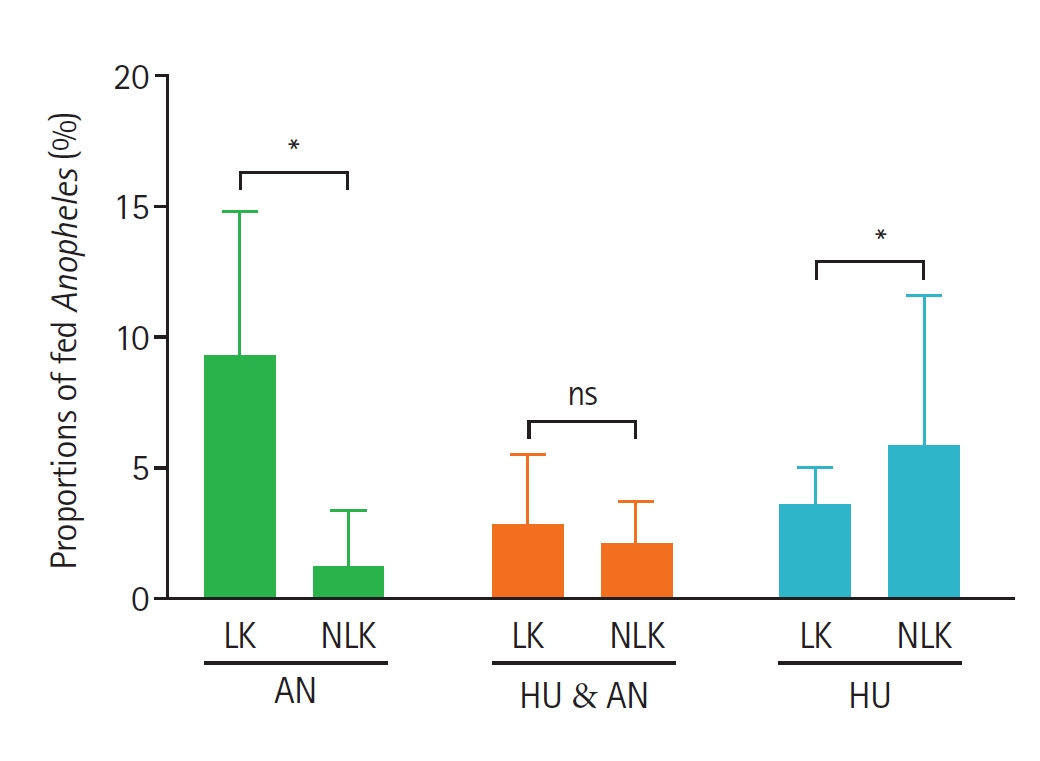
Fig. 5.Proportions of fed Anopheles mosquitoes based on the identified blood-meal sources and distance of animal corrals from human dwellings. Bars show the mean±SE proportions of blood-fed Anopheles collected from corrals located at less than 12 meters and those at 12 meters and above from human dwellings. Animal host (AN), human host (HU), and mixed blood meal index of human and animal hosts (HU & AN). Asterisks indicate statistically significant differences (P<0.05), and “ns” denotes a non-significant difference between the 2 household groups by Mann–Whitney test at 95% confidence interval.
Table 1.Morphological vs. molecular species identification
Table 1.
|
Morphological ID |
Molecular ID |
No. of specimens |
Discrepancies |
|
Anopheles gambiae s.l. |
An. arabiensis
|
446 |
Matches molecular confirmation |
|
An. gambiae s.l. |
An. gambiae s.s. |
89 |
Matches molecular confirmation |
|
An. gambiae s.l. |
Not amplified |
52 |
Potential discrepancy; retained as morphological ID |
|
An. funestus s.l. |
An. funestus s.s. |
18 |
Matches molecular confirmation |
|
An. funestus s.l. |
Not amplified |
6 |
Potential discrepancy; retained as morphological ID |
Table 2.Composition and abundance of vertebrate hosts in studied villages
Table 2.
|
Villages (n) |
Host |
|
Human |
Cow |
Sheep |
Goat |
Chicken |
Dog |
|
Old Misungwi (4,324) |
537 |
3,598 |
14 |
11 |
120 |
44 |
|
Iteja (1,295) |
238 |
610 |
175 |
210 |
52 |
10 |
|
Mwamanga (905) |
146 |
429 |
169 |
91 |
63 |
7 |
|
Mwanangwa (3,710) |
840 |
2,543 |
98 |
12 |
104 |
113 |
|
Lubuga (2,154) |
492 |
1,046 |
524 |
18 |
47 |
27 |
|
Total (12,388) |
2,253 |
8,226 |
980 |
342 |
386 |
201 |
|
Mean±SE |
451±122 |
1,645±614 |
196±87 |
68±38 |
77±15 |
40±19 |
Table 3.Abdominal status of female Anopheles mosquitoes by place of collection
Table 3.
|
Blood digestion stage (n) |
Livestock keeping households |
Non-livestock keeping households |
|
Total |
Indoor |
Outdoor |
Total |
Indoor |
Outdoor |
|
Fed (231) |
150 (64.9) |
48 (48.0) |
102 (52.0) |
81 (35.1) |
52 (64.2) |
29 (35.8) |
|
Unfed (362) |
217 (61.9) |
96 (30.3) |
121 (69.7) |
145 (39.1) |
124 (85.5) |
21 (14.5) |
|
Gravid (11) |
7 (63.6) |
2 (28.6) |
5 (71.4) |
4 (36.4) |
3 (75.0) |
1 (25.0) |
|
Half gravid (7) |
2 (28.6) |
1 (50.0) |
1 (50.0) |
5 (71.4) |
4 (80.0) |
1 (20.0) |
|
Total (611) |
376 (61.5) |
147 (32.7) |
229 (67.3) |
235 (38.5) |
183 (73.6) |
52 (26.4) |
Table 4.FR and host preference of Anopheles mosquitoes collected in Misungwi, Tanzania
Table 4.
|
Host type |
Host availability (%) |
Proportion of blood meals (%) |
Foraging ratio (BBI and HBI) |
Feeding preference |
Interpretation |
|
An. arabiensis (n=47, 57.6%) |
Humans |
18.2 |
25 |
1.38 |
Slight preference |
Opportunistic, feeds on both humans and livestock; higher tendency outdoors |
|
Cattle |
66.4 |
55 |
0.83 |
Random–avoidance |
Reflects zoophilic tendency when livestock are near houses |
|
Goats |
2.8 |
8 |
2.86 |
Strong preference |
Indicates opportunistic feeding near small ruminants |
|
Sheep |
7.9 |
7 |
0.89 |
Neutral |
No significant bias |
|
Chickens |
3.1 |
3 |
0.97 |
Neutral |
Minor feeding |
|
Dogs |
1.6 |
2 |
1.25 |
Mild preference |
Possible opportunistic feeding outdoors |
|
An. gambiae s.s. (n=31, 38.4%) |
Humans |
18.2 |
80 |
4.4 |
Strong preference |
Highly anthropophilic; main malaria vector indoors |
|
Cattle |
66.4 |
15 |
0.23 |
Avoidance |
Avoids livestock; prefers humans |
|
Goats |
2.8 |
2 |
0.71 |
Slight avoidance |
Rarely feeds on small ruminants |
|
Sheep |
7.9 |
1 |
0.13 |
Avoidance |
Prefers humans |
|
Chickens |
3.1 |
1 |
0.32 |
Avoidance |
Prefers humans |
|
Dogs |
1.6 |
1 |
0.63 |
Avoidance |
Avoids livestock; prefers humans |
|
An. funestus (n=3, 3.9%) |
Humans |
18.2 |
90 |
4.95 |
Very strong preference |
Strictly anthropophilic, indoor feeder |
|
Cattle |
66.4 |
8 |
0.12 |
Avoidance |
Minimal feeding on livestock |
|
Goats |
2.8 |
1 |
0.36 |
Avoidance |
Minimal feeding on livestock |
|
Sheep |
7.9 |
1 |
0.13 |
Avoidance |
Minimal feeding on livestock |
|
Chickens |
3.1 |
– |
– |
Avoidance |
Minimal feeding on livestock |
|
Dogs |
1.6 |
– |
– |
Avoidance |
Minimal feeding on livestock |
References
- 1. World Health Organization. World malaria report 2024. World Health Organization; 2025.
- 2. Kapfunde A. The effectiveness of indoor residual spraying program in reducing malaria incidence in Mutasa District, 2020 to 2022 [dissertation]. Africa University; 2024.
- 3. Bachan EG, Adokiya MN, Tobin-West C. Challenges of implementing malaria control interventions in resource limited countries: a systematic review. P H J Public Health 2025;1:57-69.
- 4. Takken W, Charlwood D, Lindsay SW. The behaviour of adult Anopheles gambiae, sub-Saharan Africa's principal malaria vector, and its relevance to malaria control: a review. Malar J 2024;23:161. https://doi.org/10.1186/s12936-024-04982-3
- 5. Tung GA, Fonseca DM. Internal and external drivers interact to create highly dynamic mosquito blood-feeding behaviour. Proc Biol Sci 2024;291:20241105. https://doi.org/10.1098/rspb.2024.1105
- 6. Kitole FA, Sesabo JK. Smallholder livestock keepers’ breeding choices and its implication on poverty reduction in developing countries: empirical evidence from Tanzania. Glob Soc Welf 2022;9:241-51. https://doi.org/10.1007/s40609-022-00252-9
- 7. Mramba RP, Mapunda PE. Management factors associated with the prevalence of gastrointestinal parasites in cattle: a case study of small-scale farms of the Nyamagana District in Mwanza, Tanzania. Ghana J Anim Sci 2024;14:108-19. https://doi.org/10.4314/gjansci.v14i1.11
- 8. Mwalugelo YA, Mponzi WP, Muyaga LL, et al. Livestock keeping, mosquitoes and community viewpoints: a mixed methods assessment of relationships between livestock management, malaria vector biting risk and community perspectives in rural Tanzania. Malar J 2024;23:213. https://doi.org/10.1186/s12936-024-05039-1
- 9. Kemibala EE, Mafra-Neto A, Dekker T, et al. A zooprophylaxis strategy using L-lactic acid (Abate) to divert host-seeking malaria vectors from human host to treated non-host animals. Malar J 2020;19:52. https://doi.org/10.1186/s12936-020-3136-9
- 10. Adugna T, Yewhelew D, Getu E. Bloodmeal sources and feeding behavior of anopheline mosquitoes in Bure district, northwestern Ethiopia. Parasit Vectors 2021;14:166. https://doi.org/10.1186/s13071-021-04669-7
- 11. Lardeux F, Loayza P, Bouchité B, Chavez T. Host choice and human blood index of Anopheles pseudopunctipennis in a village of the Andean valleys of Bolivia. Malar J 2007;6:8. https://doi.org/10.1186/1475-2875-6-8
- 12. Reeves LE, Burkett-Cadena ND. Mosquito blood meal analysis. Cold Spring Harb Protoc 2024;2024:pdb.top107706. https://doi.org/10.1101/pdb.top107706
- 13. National Bureau of Statistics. The 2022 population and housing census. Ministry of Finance and Planning; 2022.
- 14. Mosha JF, Kulkarni MA, Messenger LA, et al. Protocol for a four parallel-arm, single-blind, cluster-randomised trial to assess the effectiveness of three types of dual active ingredient treated nets compared to pyrethroid-only long-lasting insecticidal nets to prevent malaria transmitted by pyrethroid insecticide-resistant vector mosquitoes in Tanzania. BMJ Open 2021;11:e046664. https://doi.org/10.1136/bmjopen-2020-046664
- 15. Gillies MT, Wilkes TJ. The range of attraction of animal baits and carbon dioxide for mosquitoes: studies in a freshwater area of West Africa. Bull Entomol Res 1972;61:389-404. https://doi.org/10.1017/S0007485300047295
- 16. Takken W, Knols BG. Odor-mediated behavior of Afrotropical malaria mosquitoes. Annu Rev Entomol 1999;44:131-57. https://doi.org/10.1146/annurev.ento.44.1.131
- 17. Donnelly B, Berrang-Ford L, Ross NA, Michel P. A systematic, realist review of zooprophylaxis for malaria control. Malar J 2015;14:313. https://doi.org/10.1186/s12936-015-0822-0
- 18. Sadanandane C, Jambulingam P, Subramanian S. Role of modified CDC miniature light-traps as an alternative method for sampling adult anophelines (Diptera: Culicidae) in the National Mosquito Surveillance Programme in India. Bull Entomol Res 2004;94:55-63. https://doi.org/10.1079/ber2003281
- 19. Kakilla C, Manjurano A, Nelwin K, et al. Malaria vector species composition and entomological indices following indoor residual spraying in regions bordering Lake Victoria, Tanzania. Malar J 2020;19:383. https://doi.org/10.1186/s12936-020-03452-w
- 20. Service MW. Mosquito ecology: field sampling methods. Applied Science Publishers; 1976.
- 21. Gillies MT, Coetzee M. A supplement to the Anophelinae of Africa south of the Sahara. South African Institute for Medical Research; 1987.
- 22. Coetzee M. Key to the females of Afrotropical Anopheles mosquitoes (Diptera: Culicidae). Malar J 2020;19:70. https://doi.org/10.1186/s12936-020-3144-9
- 23. Coetzee M, Hunt RH, Wilkerson R, et al. Anopheles coluzzii and Anopheles amharicus, new members of the Anopheles gambiae complex. Zootaxa 2013;3619:246-74. https://doi.org/10.11646/zootaxa.3619.3.2
- 24. Philbert A, Lyantagaye SL, Pradel G, Ngwa CJ, Nkwengulila G. Pyrethroids and DDT tolerance of Anopheles gambiae s.l. from Sengerema District, an area of intensive pesticide usage in north-western Tanzania. Trop Med Int Health 2017;22:388-98. https://doi.org/10.1111/tmi.12850
- 25. Woyessa D, Yewhalaw D. Anopheles mosquito fauna, blood meal sources and transmission intensity from high and moderate malaria endemic areas of Ethiopia. Sci Rep 2025;15:10636. https://doi.org/10.1038/s41598-025-94739-y
- 26. Wirtz RA, Zavala F, Charoenvit Y, et al. Comparative testing of monoclonal antibodies against Plasmodium falciparum sporozoites for ELISA development. Bull World Health Organ 1987;65:39-45.
- 27. Ashine T, Kochora A, Shibru H, et al. Plasticity of blood feeding behavior of Anopheles mosquitoes in Ethiopia: a systematic review. Parasit Vectors 2024;17:408. https://doi.org/10.1186/s13071-024-06493-1
- 28. Riccetti N, Fasano A, Ferraccioli F, Gomez-Ramirez J, Stilianakis NI. Host selection and forage ratio in West Nile virus-transmitting Culex mosquitoes: challenges and knowledge gaps. PLoS Negl Trop Dis 2022;16:e0010819. https://doi.org/10.1371/journal.pntd.0010819
- 29. Rashmi P, Anupama M, Jayakrishnan TV, Jiji Joseph V. Mosquito diversity and disease transmission in Kerala: an integrated review of species ecology, habitat, and public health impact. Int J Mosq Res 2025;12:1-13. https://doi.org/10.22271/23487941.2025.v12.i2a.827
- 30. Chan K, Cano J, Massebo F, Messenger LA. Cattle-related risk factors for malaria in southwest Ethiopia: a cross-sectional study. Malar J 2022;21:179. https://doi.org/10.1186/s12936-022-04202-w
- 31. Nyangau PN, Nzuma JM, Irungu P, Kassie M. Evaluating livestock farmers knowledge, beliefs, and management of arboviral diseases in Kenya: a multivariate fractional probit approach. PLoS Negl Trop Dis 2021;15:e0009786. https://doi.org/10.1371/journal.pntd.0009786
- 32. Belay AK, Asale A, Sole CL, et al. Feeding habits and malaria parasite infection of Anopheles mosquitoes in selected agroecological areas of Northwestern Ethiopia. Parasit Vectors 2024;17:412. https://doi.org/10.1186/s13071-024-06496-y
- 33. Mayagaya VS, Nkwengulila G, Lyimo IN, et al. The impact of livestock on the abundance, resting behaviour and sporozoite rate of malaria vectors in southern Tanzania. Malar J 2015;14:17. https://doi.org/10.1186/s12936-014-0536-8
- 34. Machani MG, Ochomo E, Amimo F, et al. Behavioral responses of pyrethroid resistant and susceptible Anopheles gambiae mosquitoes to insecticide treated bed net. PLoS One 2022;17:e0266420. https://doi.org/10.1371/journal.pone.0266420
- 35. Mburu MM, Zembere K, Mzilahowa T, et al. Impact of cattle on the abundance of indoor and outdoor resting malaria vectors in southern Malawi. Malar J 2021;20:353. https://doi.org/10.1186/s12936-021-03885-x
 , Deokary J. Matiya1,2
, Deokary J. Matiya1,2 , Winifrida Kidima1
, Winifrida Kidima1 , Jaeyul Kwon3
, Jaeyul Kwon3 , Bo-In Kwon4,5,*
, Bo-In Kwon4,5,* , Ernest Mazigo6,*
, Ernest Mazigo6,*