Abstract
The Kato-Katz (KK) method is a well-known method of fecal examination for helminthiases. Its diagnostic sensitivity was found very high for clonorchiasis. The present study evaluated the correlation of Clonorchis sinensis egg counts by the KK method with those by direct smear and formalin-ether (FE) technique. The egg counts obtained by the KK method (Y) were correlated with the counts by direct smear (X) with the equation of Y = 659.4 + 0.266X (r2 = 0.738), but not with those by the FE method. The present study demonstrated that the KK method and direct smear were useful for both qualitative and quantitative diagnosis of clonorchiasis, especially in the field.
-
Key words: clonorchiasis, EPG, Kato-Katz method, direct smear
The Kato thick smear technique was originally designed for mass screening of ascariasis or other helminthiases, but is satisfactory only for qualitative diagnosis (
Beaver et al., 1984). For simultaneous qualitative and quantitative examination of helminthiasis, the Kato-Katz (KK) method was developed by modification of Kato thick smear, which uses a calibrated plastic or cardboard template to contain a consistent volume of fecal sample (
Beaver et al., 1984). The KK method had been shown to be better than Stoll's dilution egg counting technique for quantitative analysis of intestinal helminthiases (
Hong et al., 1992). Therefore, the KK method is useful for both qualitative and quantitative analysis, and is preferred for counting helminth eggs in feces, especially in the field (
Odongo-Aginya et al., 1995). Recently, we found that the KK method is a highly reliable diagnostic method for clonorchiasis compared to formalin-ether technique (FE) and direct smear (
Hong et al., 2003). The KK method showed diagnostic sensitivity of 98.9%, which was the highest rate ever recorded, and was sensitive even in lightly infected cases of clonorchiasis.
The KK method and direct smear are the only techniques that can be used for both qualitative and quantitative diagnosis of helminthiases (
Beaver et al., 1984). The combination of KK method with direct smear resulted in increase of diagnostic sensitivity, especially in the infections of
Schistosoma mansoni and hookworms (
Engels et al., 1996). However, the correlation of EPGs obtained by both methods was not described in detail.
The present study analyzed the data of our previous report (
Hong et al., 2003) to evaluate the correlation of egg counts per gram of feces (EPG) determined by the KK method with those estimated by the FE method or direct smear. Briefly, 300 residents from an endemic area of clonorchiasis in Heilongjiang, China, were screened for
C. sinensis infection by a single KK smear. An experienced technician examined each fecal specimen by reading 3 KK, 3 FE, and 6 direct smears per specimen. Among 300 subjects, 27 residents were negative for
C. sinensis eggs; therefore, EPGs of 273 people were calculated, and analyzed for the evaluation of correlations of EPGs among three stool examinations.
All three methods could count numbers of eggs of
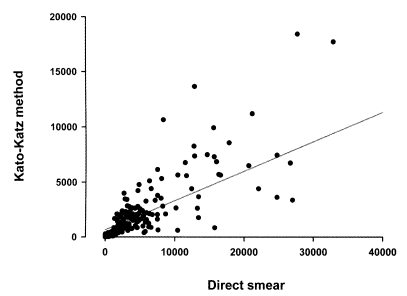
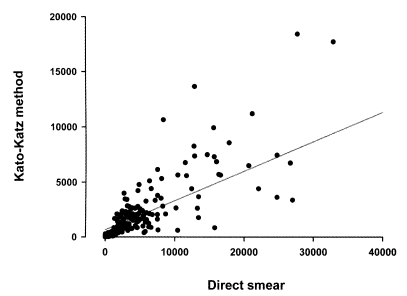
C. sinensis but the range of egg counts was variable, and became wider when the EPG counts increased. The average EPGs of 273 egg positive residents were 0-36,488 by KK method, 0.3-3,887 by FE method, and 0-181,750 by direct smear, respectively. The egg counts by the KK method (Y) were correlated with the counts by direct smear (X) with the equation of Y = 659.4 + 0.266X (r
2 = 0.738) (
Fig. 1), but not correlated with those by the FE method.
The KK method is widely used to estimate EPGs for routine quantitative diagnosis in helminthiases. It produces a consistent smear of 41.7 mg of feces, and egg counts on one smear are converted to EPG by multiplying a constant 24 (
Ebrahim et al., 1997). Due to the disadvantage of requiring a clearing time of several hours, a quick variant method has been developed, which uses a stainless steal template capable of delivering 20 mg of stools (
Peters et al., 1980). The "quick and thin" KK smear is easy to prepare specimen and make it cleared rapidly (15 min.), and the egg counts were proportional to those obtained by the classical KK smear (
Peters et al., 1980). A glass coverslip modification was also proposed to allow an immediate examination of a smear (
Teesdale and Amin, 1976a,
1976b;
Teesdale et al., 1985).
Direct smear is estimated to contain about 2 mg of feces, and EPG can be calculated by multiplying eggs per smear by 500 (
Beaver et al., 1984). Therefore, it also permits quantitative diagnosis of intestinal helminth infections. The FE method requires at least 1 g of feces for proper concentration of eggs; therefore the EPG can be estimated by reading a smear prepared by the FE method. However, concentration techniques have been known to be unreliable for egg counts due to several factors, i.e., differences in the character of the specimens or the proficiency of technicians (
Beaver et al., 1984). In the present study, the FE method can detect extremely light-infected cases more sensitively than the KK method (
Hong et al., 2003), but EPGs determined by the FE method were not reliable, and were not closely related with those by the KK or direct smears.
In the present study, it was demonstrated that the EPGs determined by KK method and by direct smear are correlated well. Therefore, both methods can be applicable for both qualitative and quantitative analysis in an endemic area of clonorchiasis, especially where the equipment for the FE method is not available.
References
- 1. Beaver PC, Jung RC, Cupp EW. Clinical parasitology. 1984. 9th ed. Philadelphia, USA. Lea & Febiger; p. 733-746.
- 2. Chai JY, Yang YT, Lee SH, Seo BS. The detectability of helminth eggs from feces by cellophane thick smear technique. Korean J Parasitol 1982;20:14-20.
- 3. Ebrahim A, El-Morshedy H, Omer E, El-Daly S, Barakat R. Evaluation of the Kato-Katz thick smear and formol ether sedimentation techniques for quantitative diagnosis of Schistosoma mansoni infection. Am J Trop Med Hyg 1997;57:706-708.
- 4. Engels D, Nahimana S, Gryseels B. Comparison of the direct faecal smear and two thick smear techniques for the diagnosis of intestinal parasitic infections. Trans R Soc Trop Med Hyg 1996;90:523-525.
- 5. Hong SJ, Woo HC, Han JH, Kim HJ. Comparative study on the effectiveness of modified Kato's cellophane thick smear and Stoll's dilution egg counting technique for quantitative fecal examination of helminth eggs. Korean J Parasitol 1992;30:141-145.
- 6. Hong ST, Choi MH, Kim CH, Chung BS, Ji Z. The Kato-Katz method is reliable for diagnosis of Clonorchis sinensis infection. Diagn Microbiol Infect Dis 2003;47:345-347.
- 7. Odongo-Aginya EI, Taylor MG, Sturrock RF, Ackers JP, Doehring E. Field evaluation of an improved Kato-Katz thick smear technique for quantitative determination of helminth eggs in faeces. Trop Med Parasitol 1995;46:275-277.
- 8. Peters PA, El Alamy M, Warren KS, Mahmoud AA. Quick Kato smear for field quantification of Schistosoma mansoni eggs. Am J Trop Med Hyg 1980;29:217-219.
- 9. Teesdale CH, Amin MA. Comparison of the Bell technique, a modified Kato thick smear technique, and a digestion method for the field diagnosis of schistosomiasis mansoni. J Helminthol 1976a;50:17-20.
- 10. Teesdale CH, Amin MA. A simple thick-smear technique for the diagnosis of Schistosoma mansoni infection. Bull WHO 1976b;54:703-705.
- 11. Teesdale CH, Fahringer K, Chitsulo L. Egg count variability and sensitivity of a thin smear technique for the diagnosis of Schistosoma mansoni. Trans R Soc Trop Med Hyg 1985;79:369-373.
Fig. 1Frequency distribution of eggs per gram of feces (EPG) determined by Kato-Katz method against EPGs by direct smear. A linear regression curve is plotted in the graph.
Citations
Citations to this article as recorded by

- Assessment of the application of the FA280—a fully automated fecal analyzer for diagnosing clonorchiasis: a mixed-method study
Si-Yue Huang, Qing-Sheng Zeng, Xin-Fu Shi, Yun-Ting He, Yue-Yi Fang, Ying-Si Lai
Infectious Diseases of Poverty.2025;[Epub] CrossRef - Performance of Mini Parasep® SF stool concentrator kit, Kato-Katz, and formalin-ethyl acetate concentration methods for diagnosis of opisthorchiasis in Northeast Thailand
Kulthida Y. Kopolrat, Seri Singthong, Narong Khuntikeo, Watcharin Loilome, Chanika Worasith, Chutima Homwong, Chompunoot Wangboon, Patiwat Yasaka, Chatanun Eamudomkarn, Opal Pitaksakulrat, Krisnakorn Tonkhamhak, Arunee Paeyo, Thomas Crellen, Jiraporn Sith
Parasites & Vectors.2022;[Epub] CrossRef - Clonorchis sinensis and clonorchiasis
Byoung-Kuk Na, Jhang Ho Pak, Sung-Jong Hong
Acta Tropica.2020; 203: 105309. CrossRef - Improving diagnostic performance of the Kato-Katz method for Clonorchis sinensis infection through multiple samples
Men-Bao Qian, Shi-Feng Zhuang, Shi-Qiao Zhu, Xiao-Mao Deng, Zheng-Xiang Li, Xiao-Nong Zhou
Parasites & Vectors.2019;[Epub] CrossRef - Performance evaluation of existing immunoassays for Clonorchis sinensis infection in China
Hong-Mei Li, Men-Bao Qian, Yi-Chao Yang, Zhi-Hua Jiang, Kang Wei, Jia-Xu Chen, Jun-Hu Chen, Ying-Dan Chen, Xiao-Nong Zhou
Parasites & Vectors.2018;[Epub] CrossRef - Current status and perspectives of Clonorchis sinensis and clonorchiasis: epidemiology, pathogenesis, omics, prevention and control
Ze-Li Tang, Yan Huang, Xin-Bing Yu
Infectious Diseases of Poverty.2016;[Epub] CrossRef - Characterization of immunogenic Clonorchis sinensis protein fractions by gel filtration chromatography
Duan Pham Ngoc, Yuji Arimatsu, Sasithorn Kaewkes, Banchob Sripa
Asian Pacific Journal of Tropical Disease.2015; 5(4): 284. CrossRef - Eosinophilia in Infectious Diseases
Elise M. O’Connell, Thomas B. Nutman
Immunology and Allergy Clinics of North America.2015; 35(3): 493. CrossRef - Evaluation of Kato-Katz and spontaneous sedimentation methods for the diagnosis of platynosomiasis in Neotropical primates
Vitor Luís Tenório Mati, Hudson Alves Pinto, Alan Lane de Melo
Revista Brasileira de Parasitologia Veterinária.2015; 24(1): 108. CrossRef - Microscopic Examination of Gallbladder Stones Improves Rate of Detection of Clonorchis sinensis Infection
Tie Qiao, Rui-hong Ma, Xiao-bing Luo, Pei-ming Zheng, Zhen-liang Luo, Liu-qing Yang
Journal of Clinical Microbiology.2013; 51(8): 2551. CrossRef - Effect of Control Strategies on Prevalence, Incidence and Re-infection of Clonorchiasis in Endemic Areas of China
Min-Ho Choi, Sue K. Park, Zhimin Li, Zhuo Ji, Gui Yu, Zheng Feng, Longqi Xu, Seung-Yull Cho, Han-Jong Rim, Soon-Hyung Lee, Sung-Tae Hong, Banchob Sripa
PLoS Neglected Tropical Diseases.2010; 4(2): e601. CrossRef - Changes in Sonographic Findings after Treatment of Patients with Clonorchiasis in a Heavy Endemic Area
Dongil Choi, Yong Hwan Jeon, Geun-Chan Lee, Min-Ho Choi, Sung-Tae Hong
The Korean Journal of Parasitology.2009; 47(1): 19. CrossRef - Prevalence of clonorchiasis in patients with gastrointestinal disease: A Korean nationwide multicenter survey
Ho Gak Kim, Jimin Han, Myung-Hwan Kim, Kyu Hyun Cho, Im Hee Shin, Gwang Ha Kim, Jae Seon Kim, Jin Bong Kim, Tae Nyeun Kim, Tae Hyeon Kim, Tae Hyo Kim, Jae Woo Kim, Ji Kon Ryu, Young-Soo Moon, Jong Ho Moon, Sung Jae Park, Chan Guk Park, Sung-Jo Bang, Chang
World Journal of Gastroenterology.2009; 15(1): 86. CrossRef - Evaluation of techniques for detection of small trematode eggs in faeces of domestic animals
Nguyen Thi Lan Anh, Nguyen Thi Phuong, Giang Hoang Ha, Luong To Thu, Maria Vang Johansen, Darwin K. Murrell, Stig Milan Thamsborg
Veterinary Parasitology.2008; 156(3-4): 346. CrossRef