Abstract
The 27 kDa cathepsin L-like cysteine protease of Spirometra erinacei plerocercoid is known to play an important function in tissue penetration, nutrient uptake and immune modulation in human sparganosis. In the present study, the expression of this enzyme was examined at different developmental stages of S. erinacei including immature egg, coracidium, plerocercoid in tadpole and rat, and adult. Proteolytic activity against carboxybenzoyl-phenylalanyl-arginyl-7-amino-4-methylcoumarin was detected in the extracts of coracidia and plerocercoid while no activity was observed in those of immature egg and adult. The specific activity in coracidial extracts was lower than that in the plerocercoid. Reverse transcription-polymerase chain reaction and Northern blot analysis demonstrated that the gene was expressed in the coracidium and plerocercoid but not in immature egg and adult. These results suggest that the 27 kDa cysteine protease is only expressed in the stages involving active migration of the parasite in the host tissue.
-
Key words: Spirometra erinacei, cysteine protease, developmental expression
Spirometra erinacei is an intestinal cestode parasite found in cats and dogs. Its life cycle stages include eggs, coracidium, procercoid in crustaceans, plerocercoid (sparganum) in terrestrial vertebrates and adults in carnivorous mammals (
Lee et al., 1990;
Cho, 1996). This cestode is regarded as one of important human-infecting parasites because human sparganosis occurs worldwide especially in East and Southeast Asian countries. Once infected, sparganum migrates through the tissues, forming a tortuous track and granuloma around it; sometimes the larva invades human brain and causes several neurological symptoms (Chang et al., 1992).
Migrating ability of sparganum through the host tissue has long been considered to be associated with secreted proteolytic enzymes. Presently, three species of serine proteases (
Kong et al., 1994a) and three species of cysteine proteases have been elucidated in the sparganum (
Fukase et al., 1985;
Song et al. 1993;
Kong et al., 1997). Of these, 27 kDa cathepsin L-like enzyme has been found to be most important in tissue invasion and nutrient uptake and its biochemical and structural nature has well been characterized (
Kong et al., 1994b;
Liu et al., 1996). The cysteine proteases also modulate host immune response by cleaving immunoglobulins or by provoking IgE antibody responses. In this study, we demonstrated that expression of the gene encoding 27 kDa cathepsin L-like cysteine protease is stage-specifically regulated.
Sparganum was harvested from naturally infected snake,
Elaphe rufodorsata and used to experimentally infect the dog. Adult
S. erinacei were collected from the dog which was orally infected with two sparganum and was allowed to grow for two months. Two weeks after the experimental infection, immature eggs of
S. erinacei were collected from dog stool. The purified immature eggs were used for either the protein extraction or coracidial hatching. The coracidium was obtained by hatching the eggs in a 29℃ incubator (
Lee et al., 1990). Procercoid larva derived from coracidium-infected freshwater copepodes,
Mesocyclops leuckarti and
Eucyclops serrulatus were used to infect tadpoles and rats. Plerocercoids were harvested from either hosts at three weeks after infection. All stages of
S. erinacei were stored in liquid nitrogen or in -70℃ deep freezer until RNA preparation or protein extraction.
Semi-quantitative reverse transcription-polymerase chain reaction (RT-PCR) was performed to compare the mRNA expression level of the 27 kDa cysteine protease gene. Total RNA samples isolated from immature egg, coracidium, sparganum and adult were quantified by spectrophotometer at 260 nm and by DNA Dipstick (Invitrogen, Carlsbad, CA, USA). Two microgram total RNA of each sample was reverse transcribed using oligo d(T)
15 primer and Molony murine leukemia virus (M-MuLV) reverse transcriptase (Life Technologies, Gaithersburg, USA). RT-PCR was carried out at 42℃ for 90 min. Aliquots of the resulting cDNA were subjected to PCR amplification with gene specific primers encoding the cathepsin L-like cysteine protease of sparganum; sense, 5'-CTGAAAGTGAGACGTACGTC-3' (SeCp70) and antisense, 5'-CAGCTGCAGTCCATCAACTG-3' (SeCp541) (D63670,
Liu et al., 1996). The cycling parameters for PCR were 94℃ for 2 min, followed by 35 cycles of 94℃ for 45 sec, 58℃ for 45 sec and 72.5℃ for 1 min with a final incubation at 72.5℃ for 10 min. The PCR condition was kept identical in all reaction. The PCR products were analysed on 1.3% agarose gel. The test was repeated three times to confirm the expression of the cDNA. Northern blot analysis was also performed with the use of
32P-labeled full-length cDNA. The full-length cDNA was obtained by PCR cloning. Ten microgram each of total RNAs of sparganum (each from frog and rat) and adult were separated on 1.2% formaldehyde agarose gel and transferred to Hybond N
+ membrane (Amersham-Pharmacia Biotech, Sweden) by capillary action. Labeling of the probe and detection of hybridization signal were carried out using
32P-labeled Rediprime DNA Labelling kit under conditions recommended by the manufacturer (Amersham-Pharmacia Biotech). Hybridization was done with 50% formamide/6× SSC solution for 16 hr at 50℃. After washing with high stringency, the membrane was autoradiographed (
Sambrook et al., 1989).
The proteolytic activities of cysteine protease in the crude extracts of immature egg, coracidium, sparganum and adult were assayed fluorometrically using synthetic substrate, carboxybenzoyl-phenylalanyl-arginyl-7-amino-4-methylcoumarin (Cbz-phe-arg- AMC, in 0.1 M sodium phosphate buffer, pH 5.7), in the presence of 5 mM dithiothreitol (DTT, Sigma, St. Louis, MO, USA). Excitation and emission wave-length was 380 nm and 460 nm, respectively. One unit of specific activity was defined as the amount of enzyme that release 1 micromole AMC per 1 hr (
Chung et al., 1997).
Modulation of the enzyme activity was observed using inhibitors acting on cysteine protease, transepoxy-succinyl-L-leucylamido(4-guanidino) butane (E-64, 10
-5 M) and iodoacetic acid (IAA, 1 mM), serine protease, aprotinin (10 µM), diisopropyl fluorophosphate (DFP, 1 mM), soybean trypsin inhibitor (SBTI, 1 mM), phenylmethyl sulfonyl fluoride (PMSF, 1 mM), N-α-p-tosyl-L-lysine chloromethylketone (TLCK, 1 mM) and L-1-tosyl-amido-2-phenyl(ethyl) chloromethylketone (TPCK, 1 mM) and metalloprotease, ethylenediaminetetraacetic acid (EDTA, 2 mM) and 1,10-phenanthroline (2 mM). All the chemicals used were from Sigma except for E-64 which was from Enzyme System Products (Livermore, CA, USA). Prior to standard assay of enzyme activity, respective inhibitors were incubated with the enzyme solution for 30 min at a room temperature (
Kong et al., 1994b).
As shown in
Table 1, the extracts from coracidia and spargana exhibited cysteine protease activity while those of immature eggs and adult worms did not. The activity appeared to be the highest in the extracts prepared from scolices of spargana, but the enzyme activity in coracidial extracts was about one fifth of that in rat sparganum.
Table 2 shows the modulation of enzyme activity by inhibitors acting on the coracidial extracts. The activity was inhibited specifically by cysteine protease inhibitors such as E-64 and IAA at the concentrations commonly used in the assays. Meanwhile, inhibitors acting on serine- and metallo-proteases such as aprotinin, DFP, SBTI, PMSF, TLCK, TPCK, EDTA and 1,10-phenanthroline did not affect the enzyme activity. Although we were not able purify the cysteine protease from the coracidia due to insufficient amount of protein available (about 250 µg), the results clearly showed the presence of cysteine protease activity in the coracidial extracts particularly, and this was confirmed with a molecular substrate that shows specificity towards cysteine protease, Cbz-Phe-Arg-AMC (
Barrett & Kirshke, 1981).
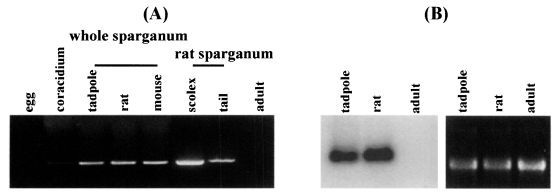
Semi-quantitative RT-PCR was carried out with the 27 kDa cysteine protease gene specific primers to examine the expression levels in immature eggs, coracidium, sparganum and adult. Expression of the 27 kDa cysteine protease was detected in the extracts of coracidia and spargana but not in those of immature eggs and adult. The RNA extracted from sparganum scolex showed higher expression level than that from tails (
Fig. 1A). Northern hybridization demonstrated that the RNA isolated from sparganum obtained from rat showed higher levels of expression than that from tadpoles. The RNA extracted from the adult worms exhibited no signals (
Fig. 1B). Despite a few inconsistencies due to varying sensitivities of the tests employed in this study, the results coincided well with the data obtained by protease assay as seen in
Table 1.
It has been speculated that the sparganum may express protective/defensive proteins due to its low host-parasite specificity (
Cho, 1996). One of the plausible mechanisms for the successful adaptation of the sparganum is its ability to express certain proteolytic enzymes. In this study, we have demonstrated that the 27 kDa cysteine protease of
S. erinacei was expressed specifically in the embryo (coracidium) and plerocercoid stages. The present result indicated that the 27 kDa cysteine protease is expressed only when penetration and active migration in the host tissues are necessary. The coracidium must penetrate the copepod gut wall to develop into the procercoid, and the plerocercoid must penetrate the gut wall of various species of vertebrate hosts to be transferred to another transport hosts. The roles and developmental modulation of cysteine proteases have been known in many helminth parasites including
Paragonimus westermani and
Haemonchus contortus (
Chung et al., 1997;
Skuce et al., 1999).
The sparganum has been shown to express cysteine proteases of different species such as 53 kDa and 21 kDa in addition to 27 kDa (
Fukase et al., 1985;
Kong et al., 1997). Further studies will be necessary to elucidate the different biological reactivities of the respective enzymes other than 27 kDa protease during the process of parasite maturation and adaptation in the various hosts.
Notes
-
This work was supported by research grant from the Korea Science and Engineering Foundation (KOSEF, 96-04-03-05-01-3).
References
- 1. Barrett AJ, Kirshke H. Cathepsin B, cathepsin H and cathepsin L. Methods Enzymol 1981;80:535-561.
- 2. Cho SY. In Weatherall DJ, Ledingham JGG, Warrell D, editors. Diphyllobothriasis and sparganosis. Oxford Textbook of Medicine. 1996. 3rd ed. Oxford, UK. Oxford Medical Publications; p. 969-970.
- 3. Chung YB, Kong Y, Yang HJ, Kang SY, Cho SY. Changes of cysteine protease activities during the maturation stages of Paragonimus westermani. J Parasitol 1997;83:902-907.
- 4. Fukase T, Matsuda A, Akihama S, Itagaki H. Purification and some properties of cysteine protease of Spirometra erinacei plerocercoid (Cestoda: Diphyllobothriidae). Jpn J Parasitol 1985;34:351-360.
- 5. Kong Y, Chung YB, Cho SY, Choi SH, Kang SY. Characterization of three neutral proteases of Spirometra mansoni plerocercoid. Parasitology 1994a;108:359-368.
- 6. Kong Y, Chung YB, Cho SY, Kang SY. Cleavage of immunoglobulin G by excretory-secretory cathepsin S-like protease of Spirometra mansoni plerocercoid. Parasitology 1994b;109:611-621.
- 7. Kong Y, Kang SY, Kim SH, Chung YB, Cho SY. A neutral cysteine protease of Spirometra mansoni plerocercoid invoking an IgE response. Parasitology 1997;114:263-271.
- 8. Lee SH, We JS, Sohn WM, Hong ST, Chai JY. Experimental life history of Spirometra erinacei. Korean J Parasitol 1990;28:163-173.
- 9. Liu DW, Kato H, Nakamura T, Sugane K. Molecular cloning and expression of the gene encoding a cysteine proteinase of Spirometra erinacei. Mol Biochem Parasitol 1996;76:11-21.
- 10. Sambrook J, Fritsch EF, Maniatis T. Molecular Cloning: A laboratory Manual. 1989. 2nd ed. New York, USA. Cold Spring Harbor Laboratory Press.
- 11. Skuce PJ, Redmond DL, Liddell S, et al. Molecular cloning and characterization of gut-derived cysteine proteinases associated with a host protective extracts from Haemonchus contortus. Parasitology 1999;119:405-412.
- 12. Song CY, Chappell CL. Purification and partial characterization of cysteine proteinase from Spirometra mansoni plerocercoids. J Parasitol 1993;79:517-524.
Fig. 1Expression of the 27 kDa cysteine protease gene according to various developmental stages of Spirometra erinacei. (A) The levels of mRNA expression observed by semi-quantitative RT-PCR analysis. The expression is detected in coracidium and sparganum while not in immature egg and adult stages. Extracts from the scolex part showing a high expression level. (B) Northern blot analysis. The RNA isolated from rat sparganum shows a slightly high expression level. The RNA extracted from the adult worms exhibits no signal.
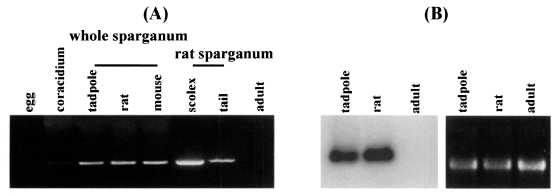
Table 1Comparison of proteolytic activity in extracts prepared variously from different developmental stages (mean ± S.D., n = 3)
Table 1
|
Enzyme source |
specific activity (units/mg protein) |
|
Egg |
0 |
|
Coracidium |
28.5 ± 3.7 |
|
Sparganum |
|
|
tadpole |
123.0 ± 7.5 |
|
rat |
134.8 ± 14.4 |
|
Sparganum (rat) |
|
|
scolex |
148.7 ± 13.5 |
|
tail |
103.5 ± 7.9 |
|
Adult |
0 |
Table 2Modulation of coracidial cysteine protease activity by various effectors (mean ± SD, n = 3)
Table 2
|
Effector |
Final conc. |
Activity (units/mg) |
Residual activity (%) |
|
Control |
5 mM DTT |
23.5 ± 3.5 |
100 |
|
Without DTT |
|
5.0 ± 1.2 |
21.3 |
|
E-64 |
10–5 M |
1.9 ± 0.4 |
8.1 |
|
IAA |
1 mM |
1.7 ± 0.3 |
7.2 |
|
Aprotinin |
10 μM |
22.3 ± 3.9 |
94.9 |
|
DFP |
1 mM |
18.4 ± 2.2 |
78.3 |
|
SBTI |
1 mM |
20.4 ± 2.8 |
86.7 |
|
PMSF |
1 mM |
16.5 ± 1.5 |
70.2 |
|
TLCK |
1 mM |
27.5 ± 2.5 |
117.0 |
|
TPCK |
1 mM |
24.3 ± 3.1 |
103.4 |
|
EDTA |
2 mM |
17.3 ± 2.4 |
73.6 |
|
1,10-phenanthroline |
2 mM |
16.6 ± 1.8 |
70.6 |
Citations
Citations to this article as recorded by

- Surgical treatment of a patient with live intracranial sparganosis for 17 years
Jialing Hu, Kaili Liao, Xiaojin Feng, Danling Jiang, Hailin Liu, Qingcui Zheng, Hai Qiu, Fuzhou Hua, Guohai Xu, Chunhua Xu
BMC Infectious Diseases.2022;[Epub] CrossRef - Follow-up study of high-dose praziquantel therapy for cerebral sparganosis
Peng Zhang, Yang Zou, Feng-Xia Yu, Zheng Wang, Han Lv, Xue-Huan Liu, He-Yu Ding, Ting-Ting Zhang, Peng-Fei Zhao, Hong-Xia Yin, Zheng-Han Yang, Zhen-Chang Wang, Siddhartha Mahanty
PLOS Neglected Tropical Diseases.2019; 13(1): e0007018. CrossRef - A cysteine protease from Spirometra erinaceieuropaei plerocercoid is a critical factor for host tissue invasion and migration
Daigo Tsubokawa, Takeshi Hatta, Hiroki Maeda, Fusako Mikami, Yukinobu Goso, Takeshi Nakamura, M. Abdul Alim, Naotoshi Tsuji
Acta Tropica.2017; 167: 99. CrossRef - Characterization of Spirometra erinaceieuropaei Plerocercoid Cysteine Protease and Potential Application for Serodiagnosis of Sparganosis
Li Na Liu, Zhong Quan Wang, Xi Zhang, Peng Jiang, Xin Qi, Ruo Dan Liu, Zi Fang Zhang, Jing Cui, Xiao-Nong Zhou
PLOS Neglected Tropical Diseases.2015; 9(6): e0003807. CrossRef - Diagnosis and stereotactic aspiration treatment of cerebral sparganosis: summary of 11 cases
Lei Deng, Pengju Xiong, Suokai Qian
Journal of Neurosurgery.2011; 114(5): 1421. CrossRef - Identification of host immune regulation candidate genes of Toxascaris leonina by expression sequenced tags (ESTs) analysis
Min Kyoung Cho, Keun Hee Lee, Sun Joo Lee, Se Won Kang, Mee Sun Ock, Yeon Chul Hong, Yong Seok Lee, Hak Sun Yu
Veterinary Parasitology.2009; 164(2-4): 242. CrossRef - Comparison of carbohydrate moieties of sparganum proteins of the snake, mouse and those of adult worm
Hyun Jong Yang
The Korean Journal of Parasitology.2003; 41(2): 135. CrossRef