Abstract
A total of 1,708 small mammals (1,617 rodents and 91 soricomorphs), including Apodemus agrarius (n = 1,400), Microtus fortis (167), Crocidura lasiura (91), Mus musculus (32), Myodes (= Eothenomys) regulus (9), Micromys minutus (6), and Tscherskia (= Cricetulus) triton (3), were live-trapped at US/Republic of Korea (ROK) military training sites near the demilitarized zone (DMZ) of Paju, Pocheon, and Yeoncheon, Gyeonggi Province from December 2004 to December 2009. Small mammals were examined for their intestinal nematodes by necropsy. A total of 1,617 rodents (100%) and 91 (100%) soricomorphs were infected with at least 1 nematode species, including Nippostrongylus brasiliensis, Heligmosomoides polygyrus, Syphacia obvelata, Heterakis spumosa, Protospirura muris, Capillaria spp., Trichuris muris, Rictularia affinis, and an unidentified species. N. brasiliensis was the most common species infecting small mammals (1,060; 62.1%) followed by H. polygyrus (617; 36.1%), S. obvelata (370; 21.7%), H. spumosa (314; 18.4%), P. muris (123; 7.2%), and Capillaria spp. (59; 3.5%). Low infection rates (0.1-0.8%) were observed for T. muris, R. affinis, and an unidentified species. The number of recovered worms was highest for N. brasiliensis (21,623 worms; mean 20.4 worms/infected specimen) followed by S. obvelata (9,235; 25.0 worms), H. polygyrus (4,122; 6.7 worms), and H. spumosa (1,160; 3.7 worms). A. agrarius demonstrated the highest prevalence for N. brasiliensis (70.9%), followed by M. minutus (50.0%), T. triton (33.3%), M. fortis (28.1%), M. musculus (15.6%), C. lasiura (13.2%), and M. regulus (0%). This is the first report of nematode infections in small mammals captured near the DMZ in ROK.
-
Key words: Nippostrongylus brasiliensis, Heligmosomoides polygyrus, Syphacia obvelata, Heterakis spumosa, Protospirura muris, Capillaria spp., Trichuris muris, Rictularia affinis, nematode, rodent, insectivore
The demilitarized zone (DMZ) is a 4-km wide boundary on the Korean Peninsula that serves as a buffer zone between South Korea (Republic of Korea; ROK) and North Korea [
1]. The DMZ consists of unmanaged lands, except for 1 village (Daeseong-dong), and is an ecologically unique and highly conserved region [
2]. As a result, there have been few reports of intestinal parasites present in wild animals near the DMZ. In addition, US/ROK-operated military training sites consist of small to large expanses of unmanaged lands similar to parts of the DMZ where wild small to large animals are often abundant.
A rodent-borne disease surveillance program, including intestinal parasite surveys, was conducted at US/ROK-operated training sites near the DMZ, Gyeonggi Province, ROK, where trematode infections were previously reported [
3-
5]. In previous studies, the striped field mouse,
Apodemus agrarius, and the Ussuri white-toothed shrew,
Crocidura lasiura, were identified as natural definitive hosts for
Plagiorchis muris [
3], in addition to several species of echinostome flukes [
4]. Moreover,
A. agrarius was identified as a natural definitive host for a human-infecting trematode,
Neodiplostomum seoulense [
5]. The present report focuses on intestinal nematode infections among 6 species of rodents (
A. agrarius,
Tscherskia triton,
Myodes regulus,
Microtus fortis,
Micromys minutus, and
Mus musculus) and 1 species of soricomorphs (
C. lasiura) live-captured near the DMZ from December 2004 to March in 2009.
A total of 1,617 rodents (6 species) and 91 soricomorphs (1 species) were live-captured, using the collapsible Sherman
® traps (3”×3.5”×9” folding traps, H.B. Sherman, Tallahassee, Florida, USA), from 3 US/ROK military training sites in Gyeonggi Province (Paju-si, Pocheon-si, and Yeoncheon-gun) located within 3-10 km from the DMZ. The captured mammals included
A. agrarius (n=1,400),
M. fortis (167),
C. lasiura (91),
M. musculus (32),
M. regulus (9),
M. minutus (6), and
T. triton (3). Trapping was conducted quarterly from December 2004 to September 2005 and from March to December 2009. Captured specimens were euthanized in accordance with a Korea University Institutional Animal Care and User Committee (IACUC) protocol under biosafety level 2 (BSL-2) laboratory conditions. The stomach, small intestine, and large intestine to the end of the rectum, were removed and placed in 50-ml glass screw-top vials in 70% alcohol until examination. The gastrointestinal content was removed and examined for intestinal helminths under a stereomicroscope [
3-
5]. Nematodes were isolated and fixed with 70% alcohol. Selected specimens were cleared in lactophenol and placed on a microscope slide with coverslip and identified using a research microscope [
6].
Of a total of 1,617 rodents and 91 soricomorphs examined, 1,048 (64.8%) and 12 (13.2%), respectively, were infected with
Nippostrongylus brasiliensis (
Table 1,
Fig. 1). In addition, rodents and soricomorphs were infected with
Heligmosomoides polygyrus (608, 37.6% and 9, 9.9%, respectively),
Syphacia obvelata (365, 22.6% and 5, 5.5%, respectively),
Heterakis spumosa (313, 19.4% and 1, 1.1%, respectively),
Protospirura muris (120, 7.4% and 3, 3.3%, respectively),
Capillaria spp. (15, 0.9% and 44, 48.4%, respectively) (
Fig. 1). and an unidentified nematode species (46, 2.8% and 13, 14.3%, respectively). Only rodents were found infected with
Rictularia affinis (13, 0.8%) and
Trichuris muris (2, 0.1%) (
Fig. 1).
A. agrarius demonstrated high nematode infection rates with 992 (70.9%) harboring
N. brasiliensis nematodes, followed by
H. polygyrus (592, 42.3%),
S. obvelata (351, 25.1%),
H. spumosa (310, 25.1%), and
Protospirura muris (120, 8.6%) (
Table 1).
M. fortis was infected with
N. brasiliensis (47, 28.1%); however, infection rates for other nematodes were relatively low (0-2.4%).
C. lasiura demonstrated the highest infection rate for
Capillaria spp. (44, 48.4%), followed by
N. brasiliensis (12, 13.2%) and
H. polygyrus (9, 9.9%).
M. musculus demonstrated the highest infection rate for
H. polygyrus (10, 31.3%), followed by
S. obvelata (8, 25.0%), and
N. brasiliensis (5, 15.6%).
N. brasiliensis was observed more frequently in
A. agrarius (70.9%), followed by
M. minutus (50.0%),
T. triton (33.3%),
M. fortis (28.1%),
M. musculus (15.6%), and
C. lasiura (13.2%).
H. polygyrus was found more frequently in
A. agrarius (42.3%), followed by
T. triton (33.3%), and
M. musculus (31.3%). The highest prevalence of
S. obvelata was observed in
M. minutus (50.0%), followed by
A. agrarius (25.1%),
M. musculus (25.0%), and
M. regulus (22.2%). The highest prevalence of
H. spumosa was observed in
A. agrarius mice (22.1%).
Capillaria spp. were observed more frequently in
C. lasiura (48.4%). The highest worm burdens were observed for
N. brasiliensis (total 21,623 worms; mean 20.4 worms/infected rodent), followed by
S. obvelata (9,235; mean 25.0),
H. polygyrus (4,122; mean 6.7),
Protospirura muris (472; mean 3.8), and
H. spumosa (1,160; mean 3.7) (
Table 1).
In the previous study, Chai et al. [
4] reported the infection status of echinostomes from
A. agrarius captured near the DMZ, ROK. They [
3,
5] also reported
Plagiorchis muris and
N. seoulense infections in
A. agrarius. However, studies of intestinal nematode infections in rodents and soricomorphs have been limited. Thus, nematode studies are needed to better understand parasite faunas in natural hosts for conserved areas, like the DMZ, and their impact on veterinary and medical health.
The results of the present study have shown that
N. brasiliensis was the most highly prevalent nematode species with the highest infection intensity among those detected in the study.
N. brasiliensis is a common and well-known intestinal nematode among rodents [
7-
9]. Its prevalence in 5 district localities was 66.7%, 80.3%, 16.2%, 71.6%, and 14.2%, respectively, for Carey Island (Klang, Malaysia), Kuala Lumpur (Malaysia), Belgrade area (Serbia), Baltimore (Maryland, USA), and Caribbean Island (Jamaica) [
8-
12]. In our study, the prevalence of
N. brasiliensis ranged from 0% (
M. regulus) to 70.9% (
A. agrarius) (overall mean, 62.1%), depending on the species of rodent or soricomorph. This figure is quite different from previous studies performed in Korea [
13-
15]. Seo et al. [
13] and Yong et al. [
14] detected high prevalences of
Nippostrongylus muris but not
N. brasiliensis among rodents from northern and northern/western parts of Korea, respectively. On the other hand, Sohn et al. [
15] detected neither
N. muris nor
N. brasiliensis in
A. agrarius mice from a southern area of Korea. The reason for this discrepancy remains to be investigated.
H. polygyrus demonstrated a high prevalence among rodents captured from various localities of the world, including Portugal, UK, and ROK (this study) and is known to enhance their susceptibility to other intestinal helminth infections [
16]. The prevalence of
H. polygyrus in our study (42.3%) was lower than that in Portugal [
16].
Heligmosomum sp.,
Heligmosomoides sp., and heligmosomid nematodes were reported from rodents in ROK [
13,
15]. Their prevalence was extremely high in rodents of southern parts of ROK (Hapcheon-gun and Gurye-gun), over 97% [
15]; however, it was comparatively lower in rodents of northern parts (Gyeonggi and Gangwon Province), 28-30% [
13].
The mean prevalence of
S. obvelata, a murine pinworm species, was 25.1% among 7 species of small mammals examined in this study. It was lower than the one in a previous study in ROK, 36.4% and 41.4%, in
R. norvegicus and
A. agrarius, respectively [
13]; however, it was higher than the other reported recently in southern regions in ROK, 5.1% [
15]. In Brazil,
Rattus rattus revealed the prevalence of 22.3%, a similar figure to our study [
17].
H. spumosa was originally reported from the cecum of a rat from Berlin, Germany, by Schneider in 1866 [
13]. In a previous study in Korea, the infection rate of
R. norvegicus,
A. agrarius, and
R. rattus (=
R. alexandrinus) with
H. spumosa was 24.2%, 6.4%, and 37.5%, respectively [
13]. In our study, its prevalence in
A. agrarius was 22.1%, much higher than the figure in a previous study [
13]. The prevalence of
H. spumosa in Serbia and Norway was 36.7% [
10] and 24.1% [
11], respectively, similar to our study.
P. muris was reported previously in the stomach of a rat from Korea in 1968 [
13]. Its overall prevalence in this study was 7.2% for all 7 small mammals examined, whereas it was 8.5% from bush rats,
Rattus fuscipes, in Australia [
18] and 5% among cotton rats,
Sigmodon hispidus, in the US [
19].
C. hepatica is common worldwide and is found in the livers of a wide variety of mammals, including humans [
20]. A previous paper from Malaysia reported the prevalence of
C. hepatica as 13.9% [
9]. In our study, we did not examine the liver but examined only the intestinal tract of the mammals. The
Capillaria spp. detected in our study may be the same species as reported from other small mammals in Korea [
13]; however, further studies are required to elucidate the species of
Capillaria. The rodent whipworm,
T. muris, is a common parasite of the cecum of house mice and rats [
21]. In
R. norvegicus, the prevalence of
T. muris was reported to be 6.0% and 14.8% in Belgrade area (Serbia) and Baltimore, Maryland (the US), respectively [
10,
11]. However, in our study, its prevalence was very low (0.1%).
The present study is significant because
S. obvelata is potentially zoonotic to cause human infections. Actually 2
S. obvelata adult specimens (females) were collected from the feces of a Bohemian child living in the Philippines [
22]. Another reason for the significance of this study is that several species of the nematodes identified in this study are used as invaluable models for human intestinal nematode infections.
N. brasiliensis is used as a model for human hookworm infections [
23],
H. polygyrus is used as a model for human ascariasis [
24], and
T. muris is used as a model for human Trichuris trichiura infection [
25].
In conclusion, 7 species of nematodes were identified, in addition to members of the genus Capillaria and an unidentified species, from the gut contents and intestinal tract of 7 species of small mammals (6 rodents and 1 soricomorph) captured near the DMZ of Gyeonggi Province, ROK. This is the first report of nematode infections among rodents and soricomorphs captured near the DMZ, ROK.
Notes
-
We have no conflict of interest related to this study.
We thank COL Hee-Choon (Sam) Lee, Chief, Force Health Protection and Preventive Medicine (FHP&PM), 65th Medical Brigade (MED BDE) and Commanders and staff of the 5th Medical Detachment, 168th Multifunctional Medical Battalion, 65th MED BDE, for their support. Funding for portions of this work was provided by the Armed Forces Health Surveillance Center- Global Emerging Infections Surveillance and Response System (AFHSC-GEIS) Operations, Silver Spring, Maryland, USA, and the Public Health Command Region-Pacific, Camp Zama, Japan.
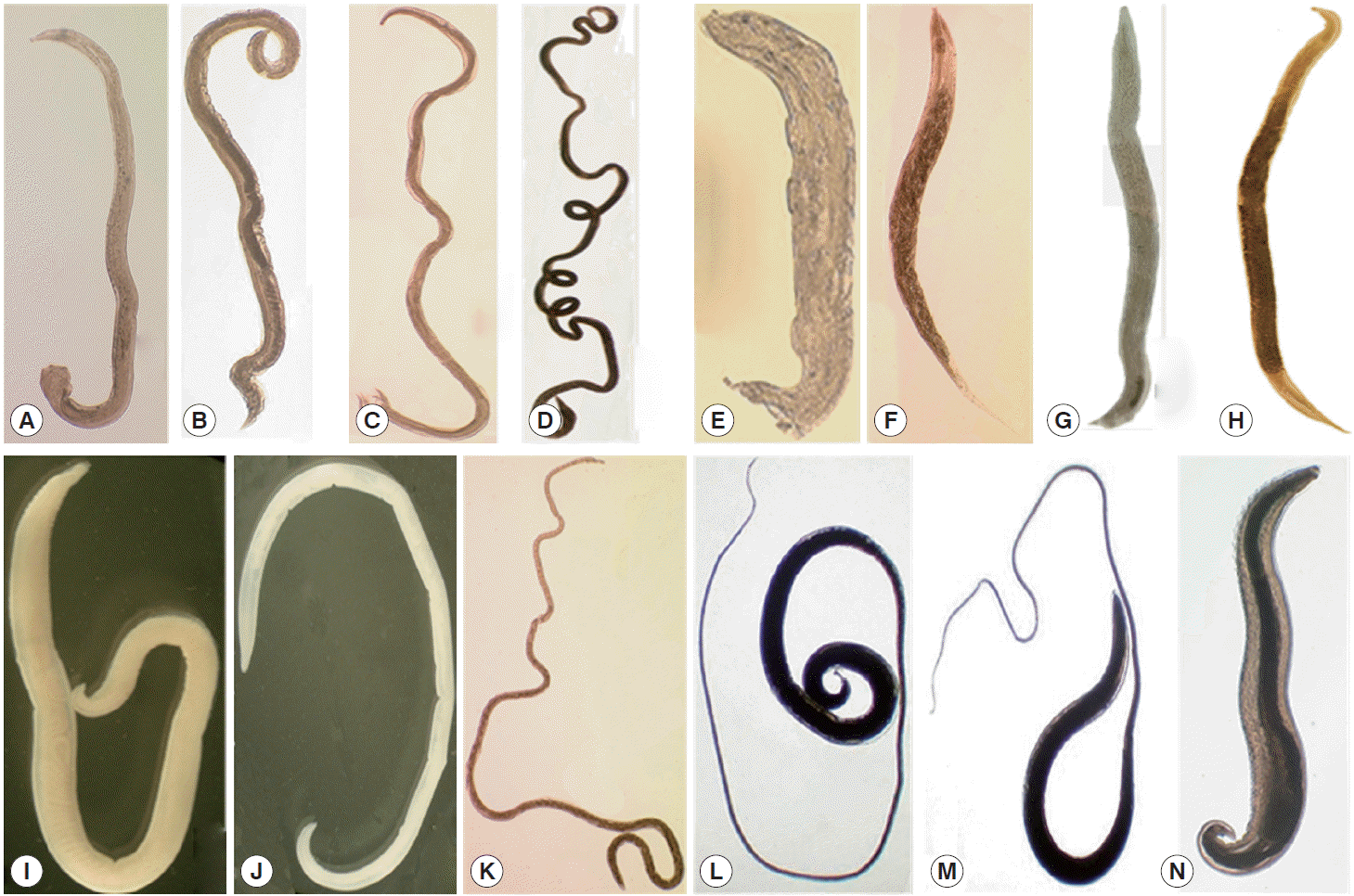
Fig. 1.Nematodes collected from wild rodents in Korea. The most popular species was Nippostrongylus brasiliensis (A, male; B, female) followed by Heligmosomoides polygyrus (C, male; D, female), Syphacia obvelata (E, male, F, female), Heterakis spumosa (G, male; H, female), Protospirura muris (I, male; J, female), Capillaria sp. (K, female), Trichuris muris (L, male; M, female), and Rictularia affinis (N, male).
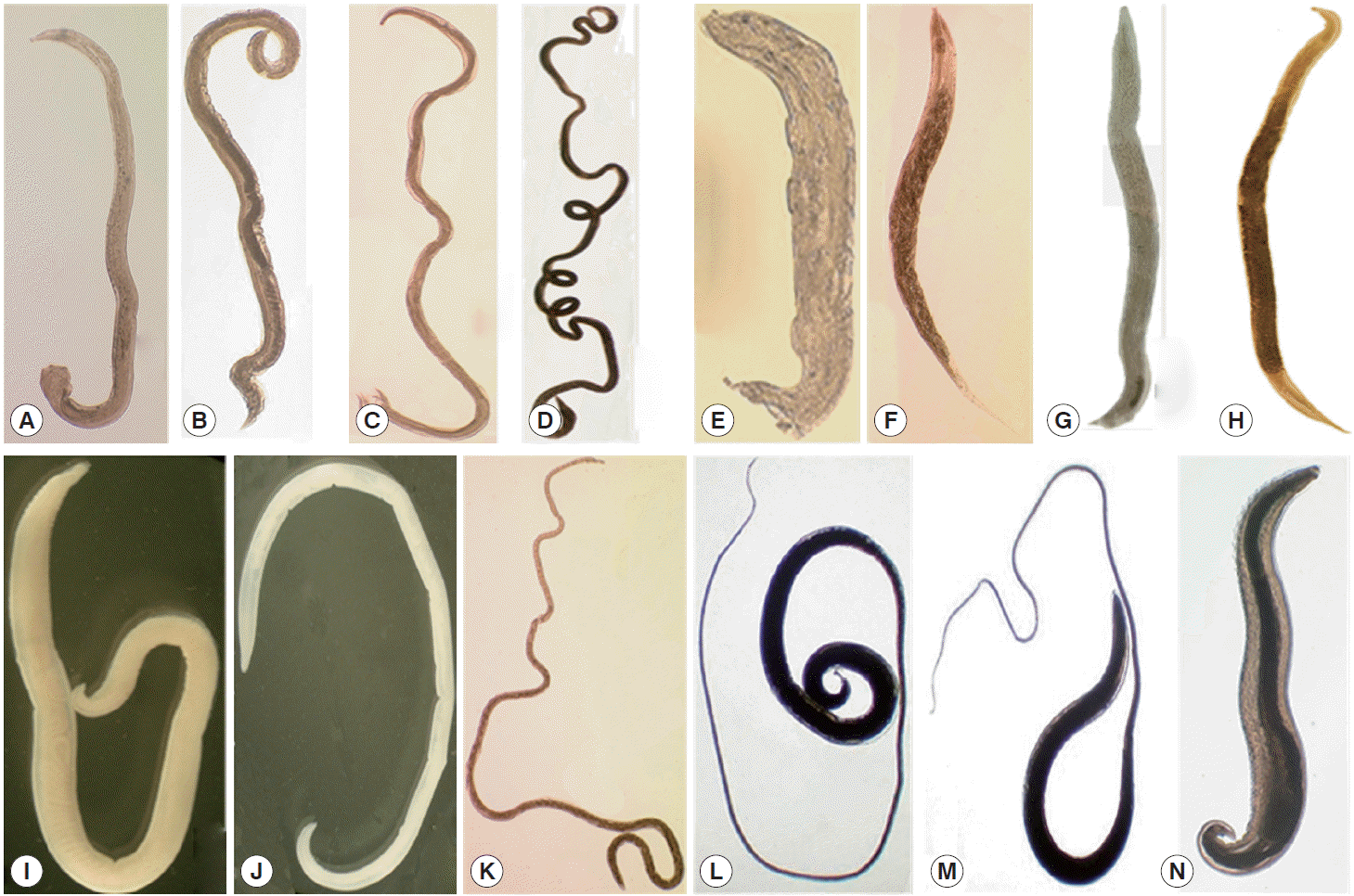
Table 1.Nematode infections in small rodents and soricomorphs captured near the DMZ of Gyeonggi Province, Korea
Table 1.
|
|
Nematodes
|
|
|
No. rodents examined |
Nippostrongyius brasiiiensis
|
Heiigmosomoides poiygyrus
|
Syphacia obvelata
|
Heterakis spumosa
|
Protospirura muris
|
Capiiiaria sp. |
Rictuiaria affinis
|
Trichuris muris
|
Others |
|
Apodemus agrarius
|
No. mice infected (%) |
1,400 |
992 (70.9) |
592 (42.3) |
351 (25.1) |
310 (22.1) |
120 (8.6) |
10 (0.7) |
13 (0.9) |
1 (0.1) |
17 (1.2) |
|
No. worms (av. no.)a
|
|
20,830 (21.0) |
3,954 (6.7) |
9,013 (25.7) |
1,146 (0.5) |
469 (3.9) |
14 (1.4) |
23 (1.8) |
1 (1.0) |
23 (1.4) |
|
Microtus fortis
|
No. mice infected (%) |
167 |
47 (28.1) |
4 (2.4) |
1 (0.6) |
0 (0.0) |
0 (0.0) |
5 (3.0) |
0 (0.0) |
0 (0.0) |
29 (17.4) |
|
No. worms (av. no.)a
|
|
698 (14.9) |
21 (5.3) |
1 (1.0) |
0 (0.0) |
0 (0.0) |
9 (1.8) |
0 (0.0) |
0 (0.0) |
211 (7.3) |
|
Crocidura iasiura
|
No. mice infected (%) |
91 |
12 (13.2) |
9 (9.9) |
5 (5.5) |
1 (1.1) |
3 (3.3) |
44 (48.4) |
0 (0.0) |
0 (0.0) |
13 (14.3) |
|
No. worms (av. no.)a
|
|
77 (6.4) |
86 (9.6) |
11 (2.2) |
2 (2.0) |
3(1.0) |
308 (7.0) |
0 (0.0) |
0 (0.0) |
21 (1.6) |
|
Mus muscuius
|
No. mice infected (%) |
32 |
5 (15.6) |
10 (31.3) |
8 (25.0) |
2 (6.3) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
1 (3.1) |
0 (0.0) |
|
No. worms (av. no.)a
|
|
9 (1.8) |
59 (5.9) |
131 (16.4) |
3 (1.5) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
3 (3.0) |
0 (0.0) |
|
Myodes reguius
|
No. mice infected (%) |
9 |
0 (0.0w) |
1 (11.1) |
2 (22.2) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
|
No. worms (av. no.)a
|
|
0 (0.0) |
1 (1.0) |
32 (16.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
|
Micromys minutus
|
No. mice infected (%) |
6 |
3 (50.0) |
0 (0.0) |
3 (50.0) |
1 (16.7) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
|
No. worms (av. no.)a
|
|
8 (2.7) |
0 (0.0) |
47 (15.7) |
9 (9.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
|
Tscherskia triton
|
No. mice infected (%) |
3 |
1 (33.3) |
1 (33.3) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
|
No. worms (av. no.)a
|
|
1 (1.0) |
1 (1.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
|
Total |
No. mice infected (%) |
1,708 |
1,060 (62.1) |
617 (36.1) |
370 (21.7) |
314 (18.4) |
123 (7.2) |
59 (3.5) |
13 (0.8) |
2 (0.1) |
59 (3.5) |
|
No. worms (av. no.)a
|
|
21,623 (20.4) |
4,122 (6.7) |
9,235 (25.0) |
1,160 (3.7) |
472 (3.8) |
331 (5.6) |
23 (1.8) |
4 (2.0) |
255 (4.3) |
References
- 1. Yoon BI, Kim HC, Kim JT. Lung worm (Metastrongylus elongatus) infection in wild boars (Sus scrofa) of the demilitarized zone, Korea. J Wildl Dis 2010;46:1052-1054.
- 2. Kim KC. Preserving biodiversity in Korea’s demilitarized zone. Science 1997;278:242-243.
- 3. Chai JY, Park JH, Guk SM, Kim JL, Kim HJ, Kim WH, Shin EH, Klein TA, Kim HC, Chong ST, Song JW, Baek LJ. Plagiorchis muris infection in Apodemus agrarius from northern Gyeonggi-do (province) near the demilitarized zone. Korean J Parasitol 2007;45:153-156.
- 4. Chai JY, Park JH, Jung BK, Guk SM, Kim JL, Shin EH, Klein TA, Kim HC, Chong ST, Baek LJ, Song JW. Echinostome infections in the striped-field mouse, Apodemus agrarius, and the Ussuri white-toothed shrew, Crocidura lasiura, caught near the demilitarized zone, Gyeonggi-do (Province), Republic of Korea. Korean J Parasitol 2009;47:311-314.
- 5. Chai JY, Park JH, Guk SM, Kim JL, Kim HJ, Kim WH, Shin EH, Klein TA, Kim HC, Chong ST, Song JW, Baek LJ. Apodemus agrarius as a new definitive host for Neodiplostomum seoulense. Korean J Parasitol 2007;45:157-161.
- 6. Beaver PC, Jung RC, Cupp EW. Clinical Parasitology. 9th ed. Philadelphia, USA. Lea & Febiger; 1984. p. 733-758.
- 7. Haley AJ. Biology of the rat nematode Nippostrongylus brasiliensis (Travassos, 1914). I. Systematics, hosts and geographic distribution. J Parasitol 1961;47:727-732.
- 8. Nursyazana MT, Mohdzain SN, Jeffery J. Biodiversity and macroparasitic distribution of the wild rat population of Carey Island, Klang. Trop Biomed 2013;30:199-210.
- 9. Siti Shafiyyah CO, Jamaiah I, Rohela M, Lau YL, Siti Aminah F. Prevalence of intestinal and blood parasites among wild rats in Kuala Lumpur, Malaysia. Trop Biomed 2012;29:544-550.
- 10. Kataranovski M, Mirkov I, Belij S, Popov A, Petrovic Z, Gaci Z, Kataranovski D. Intestinal helminths infection of rats (Ratus norvegicus) in the Belgrade area (Serbia): the effect of sex, age and habitat. Parasite 2011;18:189-196.
- 11. Easterbrook JD, Kaplan JB, Glass GE, Watson J, Klein SL. A survey of rodent-borne pathogens carried by wild-caught Norway rats: a potential threat to laboratory rodent colonies. Lab Anim 2008;42:92-98.
- 12. Waugh CA, Lindo JF, Foronda P, Angeles-Santana M, Lorenzo-Morales J, Robinson RD. Population distribution and zoonotic potential of gastrointestinal helminths of wild rats Rattus rattus and R. norvegicus from Jamaica. J Parasitol 2006;92:1014-1018.
- 13. Seo BS, Rim HJ, Yoon JJ, Koo BY, Hong NT. Studies on the parasitic helminths of Korea. III. Nematodes and cestodes of rodents. Korean J Parasitol 1968;6:123-131.
- 14. Yong TS, Chung KH, Ree HI. Infection status of intestinal parasites of field rodents in Korea. Yonsei Rep Trop Med 1991;22:55-59.
- 15. Sohn WM, Na BK, Song HJ, Kim CM, Nam GJ. Intestinal helminthic infections in striped field mice, Apodemus agrarius, from two southern regions of Korea. Korean J Parasitol 2014;52:419-423.
- 16. Behnke JM, Eira C, Rogan M, Gilbert FS, Torres J, Miquel J, Lewis JW. Helminth species richness in wild wood mice, Apodemus sylvaticus, is enhanced by the presence of the intestinal nematode Heligmosomoides polygyrus. Parasitology 2009;136:793-804.
- 17. de Araujo EO, de Moura Mendes M, Langone PQ, Müller G. The helminth parasites of Rattus rattus (Linnaeus, 1758) of urban, intermediate and rural environments in southern Brazil. Neotrop Helminthol 2014;8:19-22.
- 18. Obendorf DL. The helminth parasites of Rattus fuscipes (Waterhouse) from Victoria, including description of two new nematode species. Aust J Zool 1979;27:867-879.
- 19. Boggs JF, McMurry ST, Leslie DM Jr, Engle DM, Lochmiller RL. Influence of habitat modification on the community of gastrointestinal helminths of cotton rats. J Wildl Dis 1991;27:584-593.
- 20. Tung KC, Hsiao FC, Wang KS, Yang CH, Lai CH. Study of the endoparasitic fauna of commensal rats and shrews caught in traditional wet markets in Taichung City, Taiwan. J Microbiol Immunol Infect 2013;46:85-88.
- 21. Fahmy MAM. An investigation on the life cycle of Trichuris muris. Parasitology 1954;44:50-57.
- 22. Riley WA. A mouse oxyurid, Syphacia obvelata, as a parasite of man. J Parasitol 1919;6:89-93.
- 23. Ndlovu H, Darby M, Froelich M, Horsnell W, Lühder F, Hünig T, Brombacher F. Inducible deletion of CD28 prior to secondary Nippostrongylus brasiliensis infection impairs worm expulsion and recall of protective memory CD4+ T cell. PLoS Pathog 2014;10:e1003906.
- 24. Reynolds LA, Harcus Y, Smith KA, Webb LM, Hewitson JP, Ross EA, Brown S, Uematsu S, Akira S, Gray D, Gray M, MacDonald AS. MyD88 signaling inhibits protective immunity to the gastrointestinal helminth parasite Heligmosomoides polygyrus. J Immunol 2014;193:2984-2993.
- 25. Cliffe LJ, Grencis RK. The Trichuris muris system: a paradigm of resistance and susceptibility to intestinal nematode infection. Adv Parasitol 2004;57:255-307.